肠道微生物群衍生代谢物作为阿尔茨海默病代谢综合征相关线粒体和内溶酶体功能障碍调节剂的潜在作用。
IF 9.5
2区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
虽然肠道微生物群(GMB)产生的代谢物在代谢综合征下阿尔茨海默病(AD)的线粒体和内溶酶体功能障碍中的作用仍不清楚,但破译这些宿主-代谢物之间的相互作用是一项重大的公共卫生挑战。线粒体和内溶酶体网络(ELNs)的功能障碍在代谢综合征中起着至关重要的作用,并可加剧阿尔茨海默病的进展,这突出表明有必要研究它们之间的相互调控,以更好地了解阿尔茨海默病如何与代谢综合征联系在一起。同时,代谢紊乱与 GMB 组成的改变有关。最近的证据表明,GMB及其代谢物的组成变化可能与AD病理有关。本综述强调了代谢综合征介导的 AD 发病机制,重点是线粒体功能障碍、ELN 异常和 GMB 及其代谢物变化的相互关联作用。我们还讨论了 GMB 衍生代谢物(包括氨基酸、脂肪酸、其他代谢物和细胞外囊泡)在介导线粒体和 ELN 功能障碍方面的病理生理作用。最后,本综述提出了针对代谢综合征的 GMB 代谢物直接调节线粒体和 ELN 功能的 AD 治疗策略。本文章由计算机程序翻译,如有差异,请以英文原文为准。
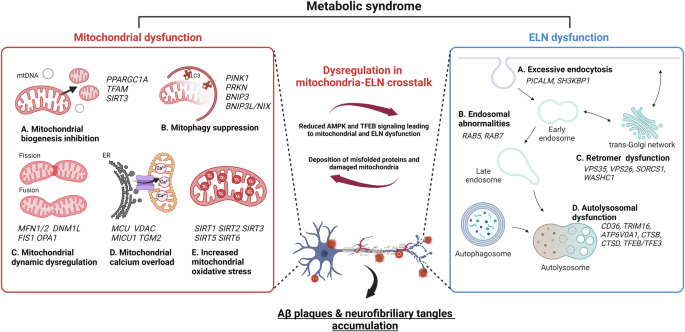
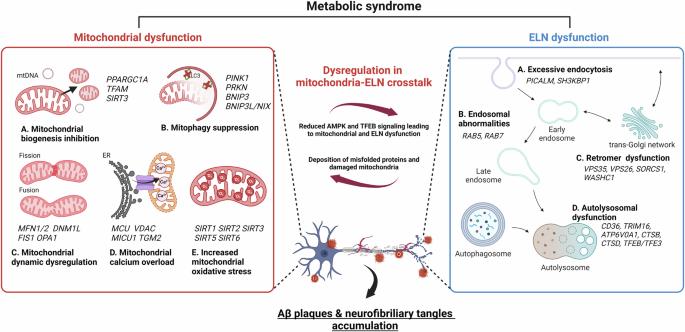
The potential role of gut microbiota-derived metabolites as regulators of metabolic syndrome-associated mitochondrial and endolysosomal dysfunction in Alzheimer’s disease
Although the role of gut microbiota (GMB)-derived metabolites in mitochondrial and endolysosomal dysfunction in Alzheimer’s disease (AD) under metabolic syndrome remains unclear, deciphering these host–metabolite interactions represents a major public health challenge. Dysfunction of mitochondria and endolysosomal networks (ELNs) plays a crucial role in metabolic syndrome and can exacerbate AD progression, highlighting the need to study their reciprocal regulation for a better understanding of how AD is linked to metabolic syndrome. Concurrently, metabolic disorders are associated with alterations in the composition of the GMB. Recent evidence suggests that changes in the composition of the GMB and its metabolites may be involved in AD pathology. This review highlights the mechanisms of metabolic syndrome-mediated AD development, focusing on the interconnected roles of mitochondrial dysfunction, ELN abnormalities, and changes in the GMB and its metabolites. We also discuss the pathophysiological role of GMB-derived metabolites, including amino acids, fatty acids, other metabolites, and extracellular vesicles, in mediating their effects on mitochondrial and ELN dysfunction. Finally, this review proposes therapeutic strategies for AD by directly modulating mitochondrial and ELN functions through targeting GMB metabolites under metabolic syndrome. Although mitochondrial and endolysosomal network (ELN) impairment in metabolic syndrome is considered a risk factor for neurodegenerative diseases, the regulatory role of gut microbiota (GMB)-derived metabolites in this dysfunction remains unclear. This research explores the roles and molecular mechanisms of mitochondrial dysfunction, ELN abnormalities, dysregulation of mitochondria-ELN crosstalk, and changes in GMB and its metabolites in metabolic syndrome, especially in relation to Alzheimer’s disease (AD). The researchers conclude by highlighting the potential of targeting GMB and its metabolites to develop new AD treatments, especially for those with metabolic syndrome. They suggest that understanding and modulating the links between gut health, mitochondrial function, and ELN activity could lead to new management strategies for AD in the context of gut microbiota and its metabolites. This summary was initially drafted using artificial intelligence, then revised and fact-checked by the author.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Experimental and Molecular Medicine
医学-生化与分子生物学
CiteScore
19.50
自引率
0.80%
发文量
166
审稿时长
3 months
期刊介绍:
Experimental & Molecular Medicine (EMM) stands as Korea's pioneering biochemistry journal, established in 1964 and rejuvenated in 1996 as an Open Access, fully peer-reviewed international journal. Dedicated to advancing translational research and showcasing recent breakthroughs in the biomedical realm, EMM invites submissions encompassing genetic, molecular, and cellular studies of human physiology and diseases. Emphasizing the correlation between experimental and translational research and enhanced clinical benefits, the journal actively encourages contributions employing specific molecular tools. Welcoming studies that bridge basic discoveries with clinical relevance, alongside articles demonstrating clear in vivo significance and novelty, Experimental & Molecular Medicine proudly serves as an open-access, online-only repository of cutting-edge medical research.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


