人类上呼吸道免疫记忆的多样性。
IF 50.5
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
摘要
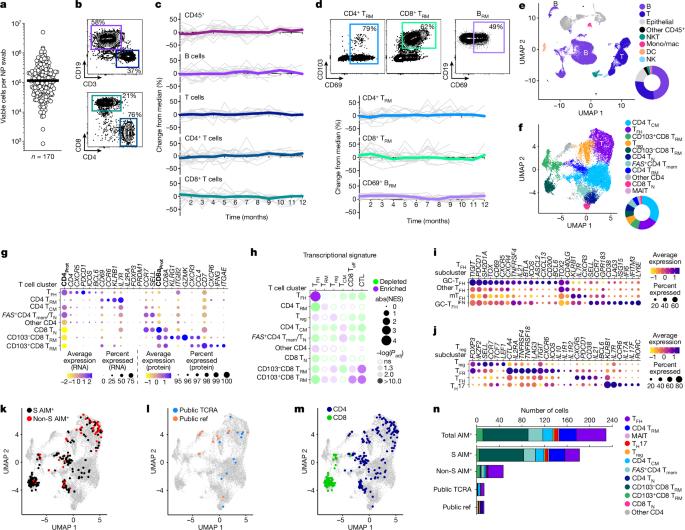
上呼吸道是一个重要的感染部位,但人们对人体上呼吸道的免疫记忆却知之甚少,这对 COVID-19 和许多其他人类疾病都有影响1-4。在这里,我们证明了鼻腔和鼻咽拭子可用于深入了解这些具有挑战性的问题,并确定了上呼吸道两个相邻解剖部位不同的免疫细胞群,包括抗原特异性记忆 B 细胞和 T 细胞。在健康成年人中,上气道免疫细胞群似乎随着时间的推移而稳定,他们每月接受一次拭子检查,时间超过 1 年,并确定了突出的组织常驻记忆 T(TRM)细胞和 B(BRM)细胞群。意想不到的是,在许多鼻咽拭子中都持续发现了生殖中心细胞。在 SARS-CoV-2 突破性感染的受试者中,鉴定出了本地病毒特异性 BRM 细胞、浆细胞和生殖中心 B 细胞,有证据表明,与血液相比,上呼吸道分区中的本地引物和 IgA+ 记忆 B 细胞更为丰富。确定了具有长寿转录特征的局部浆细胞群。确定了本地病毒特异性记忆 CD4+ TRM 细胞和 CD8+ TRM 细胞,以及多种额外的病毒特异性 T 细胞。观察到了与年龄相关的上呼吸道免疫学变化。这些发现为人类主要粘膜屏障组织的免疫记忆提供了新的认识。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Immunological memory diversity in the human upper airway
The upper airway is an important site of infection, but immune memory in the human upper airway is poorly understood, with implications for COVID-19 and many other human diseases1–4. Here we demonstrate that nasal and nasopharyngeal swabs can be used to obtain insights into these challenging problems, and define distinct immune cell populations, including antigen-specific memory B cells and T cells, in two adjacent anatomical sites in the upper airway. Upper airway immune cell populations seemed stable over time in healthy adults undergoing monthly swabs for more than 1 year, and prominent tissue resident memory T (TRM) cell and B (BRM) cell populations were defined. Unexpectedly, germinal centre cells were identified consistently in many nasopharyngeal swabs. In subjects with SARS-CoV-2 breakthrough infections, local virus-specific BRM cells, plasma cells and germinal centre B cells were identified, with evidence of local priming and an enrichment of IgA+ memory B cells in upper airway compartments compared with blood. Local plasma cell populations were identified with transcriptional profiles of longevity. Local virus-specific memory CD4+ TRM cells and CD8+ TRM cells were identified, with diverse additional virus-specific T cells. Age-dependent upper airway immunological shifts were observed. These findings provide new understanding of immune memory at a principal mucosal barrier tissue in humans. This study of immunological memory diversity in the human upper airway provides new understanding of immune memory at a major mucosal barrier tissue in humans.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature
综合性期刊-综合性期刊
CiteScore
90.00
自引率
1.20%
发文量
3652
审稿时长
3 months
期刊介绍:
Nature is a prestigious international journal that publishes peer-reviewed research in various scientific and technological fields. The selection of articles is based on criteria such as originality, importance, interdisciplinary relevance, timeliness, accessibility, elegance, and surprising conclusions. In addition to showcasing significant scientific advances, Nature delivers rapid, authoritative, insightful news, and interpretation of current and upcoming trends impacting science, scientists, and the broader public. The journal serves a dual purpose: firstly, to promptly share noteworthy scientific advances and foster discussions among scientists, and secondly, to ensure the swift dissemination of scientific results globally, emphasizing their significance for knowledge, culture, and daily life.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


