恢复耐甲氧西林金黄色葡萄球菌对β-内酰胺类抗生素的敏感性
IF 12.9
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
金黄色葡萄球菌感染历来使用β-内酰胺类抗生素治疗。然而,耐甲氧西林金黄色葡萄球菌通过获得 bla 和 mec 操作子,已不再使用这些抗生素。BlaR和/或MecR的传感器结构域检测到β-内酰胺类抗生素的存在,并将信息传递到细胞质,从而导致抗生素基因的抑制作用减弱。我们假设,抑制传感器结构域将关闭这一反应系统,从而恢复对β-内酰胺类药物的敏感性。通过对 1,100 万种化合物进行硅学搜索,我们找到了一种苯并咪唑类化合物,并最终找到了硼酸盐 4。4 的 X 射线结构与 BlaR 的活性位点丝氨酸共价结合。化合物 4 能将奥沙西林和美罗培南对耐甲氧西林金黄色葡萄球菌菌株的活性提高 16 到 4096 倍。化合物 4 与奥沙西林或美罗培南的组合在受感染的小鼠身上显示出疗效,从而验证了这一策略。本文章由计算机程序翻译,如有差异,请以英文原文为准。

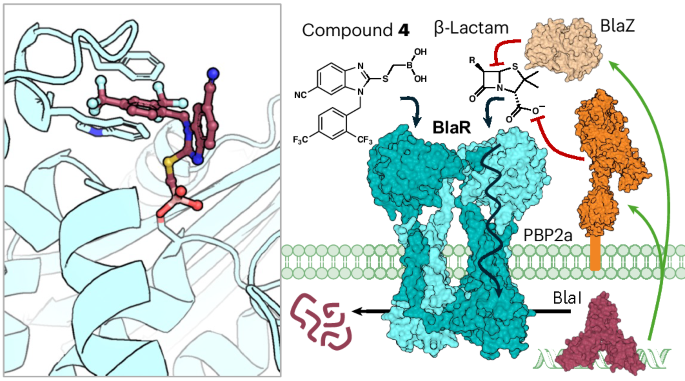
Restoring susceptibility to β-lactam antibiotics in methicillin-resistant Staphylococcus aureus
Infections by Staphylococcus aureus have been treated historically with β-lactam antibiotics. However, these antibiotics have become obsolete in methicillin-resistant S. aureus by acquisition of the bla and mec operons. The presence of the β-lactam antibiotic is detected by the sensor domains of BlaR and/or MecR, and the information is transmitted to the cytoplasm, resulting in derepression of the antibiotic-resistance genes. We hypothesized that inhibition of the sensor domain would shut down this response system, and β-lactam susceptibility would be restored. An in silico search of 11 million compounds led to a benzimidazole-based hit and, ultimately, to the boronate 4. The X-ray structure of 4 is covalently engaged with the active-site serine of BlaR. Compound 4 potentiates by 16- to 4,096-fold the activities of oxacillin and of meropenem against methicillin-resistant S. aureus strains. The combination of 4 with oxacillin or meropenem shows efficacy in infected mice, validating the strategy. β-Lactam antibiotics are detected by the sensor domain of BlaR in methicillin-resistant Staphylococcus aureus. A class of inhibitors has been developed that targets this sensor domain and prevents downstream activation of the antibiotic response pathway.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature chemical biology
生物-生化与分子生物学
CiteScore
23.90
自引率
1.40%
发文量
238
审稿时长
12 months
期刊介绍:
Nature Chemical Biology stands as an esteemed international monthly journal, offering a prominent platform for the chemical biology community to showcase top-tier original research and commentary. Operating at the crossroads of chemistry, biology, and related disciplines, chemical biology utilizes scientific ideas and approaches to comprehend and manipulate biological systems with molecular precision.
The journal embraces contributions from the growing community of chemical biologists, encompassing insights from chemists applying principles and tools to biological inquiries and biologists striving to comprehend and control molecular-level biological processes. We prioritize studies unveiling significant conceptual or practical advancements in areas where chemistry and biology intersect, emphasizing basic research, especially those reporting novel chemical or biological tools and offering profound molecular-level insights into underlying biological mechanisms.
Nature Chemical Biology also welcomes manuscripts describing applied molecular studies at the chemistry-biology interface due to the broad utility of chemical biology approaches in manipulating or engineering biological systems. Irrespective of scientific focus, we actively seek submissions that creatively blend chemistry and biology, particularly those providing substantial conceptual or methodological breakthroughs with the potential to open innovative research avenues. The journal maintains a robust and impartial review process, emphasizing thorough chemical and biological characterization.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


