高压钾银合金的结构和化学键。
IF 5.9
2区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
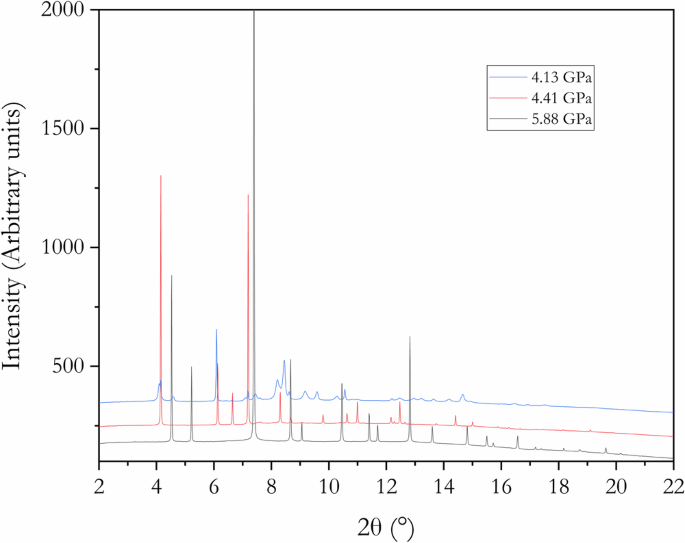
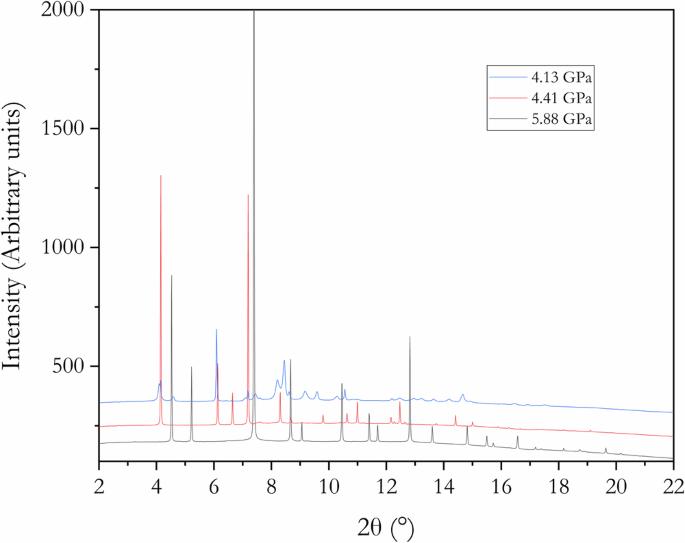
钾银合金的高压结构是压力诱导电子从电性正的钾转移到电性负的银的例子。我们利用粉末 X 射线衍射和理论计算重新研究了 KAg2、K2Ag 和 K3Ag 的晶体和电子结构。我们的研究结果在这三种物相的形态与前驱体面心立方银之间建立了联系。对于 K2Ag,我们发现了一种与 X 射线形态更匹配的无序结构。通过最大熵法获得的价电子密度分布以及电荷密度计算,让我们对这些体系中化学键的演变有了全面的了解。研究发现,K 原子在合金形成过程中共享价电子,在 K2Ag 和 KAg2 中形成了 K-Ag 和 Ag-Ag 键,而在 K3Ag 中则没有 Ag-Ag 键。这些结果表明,Zintl-Klemm 模型在描述高压二元金属间化合物的结构和键合方面可能过于简单。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Structure and chemical bonding in high-pressure potassium silver alloys
The high-pressure structures of K-Ag alloys were examples of pressure-induced electron transfer from the electropositive potassium to the electronegative silver. We re-examined the crystal and electronic structures of KAg2, K2Ag, and K3Ag using powder X-ray diffraction and theoretical calculations. Our findings establish a connection between the morphologies of these three phases and the precursor face-centered cubic Ag. For K2Ag, we discovered a disordered structure that better matches the X-ray pattern. Valence electron density distributions obtained from the maximum entropy method, along with charge density calculations, provide a comprehensive understanding of the evolution of chemical bonding in these systems. It was found that K atoms share their valence electrons during alloy formation, contributing to K-Ag and Ag-Ag bonds in K2Ag and KAg2, while no Ag-Ag bonds are present in K3Ag. These results indicate the Zintl-Klemm model may be too simplistic to describe the structure and bonding in high-pressure binary intermetallic compounds. The Zintl-Klemm concept explains the structure and chemical bonding of intermetallic compounds at high pressures — such as high-temperature superconducting metal superhydrides. Here, the authors elucidate the electronic structures of three high-pressure potassium silver alloys, providing an example of where the Zint-Klemm concept needs to be expanded.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Communications Chemistry
Chemistry-General Chemistry
CiteScore
7.70
自引率
1.70%
发文量
146
审稿时长
13 weeks
期刊介绍:
Communications Chemistry is an open access journal from Nature Research publishing high-quality research, reviews and commentary in all areas of the chemical sciences. Research papers published by the journal represent significant advances bringing new chemical insight to a specialized area of research. We also aim to provide a community forum for issues of importance to all chemists, regardless of sub-discipline.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


