直接观察致病突变体模板化蛋白质错误折叠的朊病毒式传播
IF 12.9
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
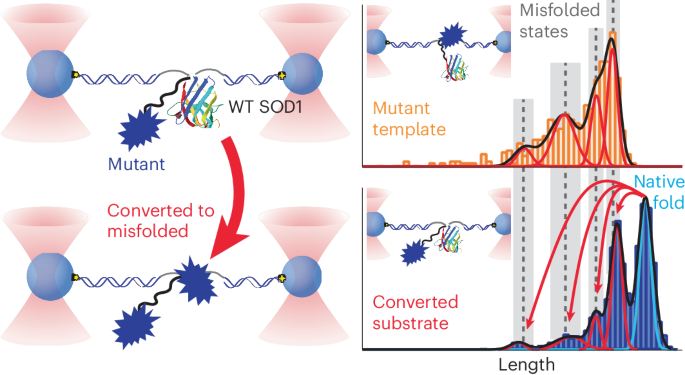
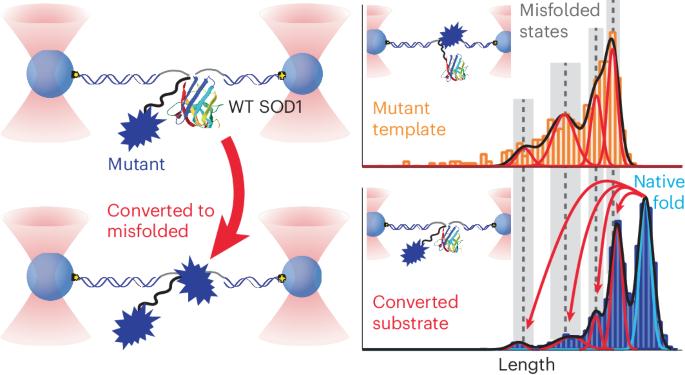
许多神经退行性疾病的特征是折叠错误的蛋白质通过原生折叠分子的模板转换而传播。然而,关于这种朊病毒样转化是如何发生的以及是什么驱动了这种转化的关键问题仍未得到解决,部分原因是技术上的挑战阻碍了对任何蛋白质转化的直接观察。我们在超氧化物歧化酶-1(SOD1)的单分子中观察到了朊病毒样转化,其错误折叠与肌萎缩侧索硬化症有关。将致病性错误折叠的 SOD1 突变体与用光学镊子夹住的野生型分子拴在一起,我们发现突变体大大增加了野生型分子的错误折叠,诱导出多种错误折叠的异构体。最重要的是,在突变体和转换后的野生型结构域中,错误折叠的模式是相同的,当错误折叠的突变体发生变化时,错误折叠的模式也会发生变化,这反映了朊病毒样转换所预期的模板效应。组合测量结果显示,随着转化的进行,系链异二聚体的酶活性降低,这反映了单分子结果。对疾病特异性表位敏感的抗体与转化后的蛋白质结合,这意味着转化产生了与疾病相关的错误折叠构象。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Direct observation of prion-like propagation of protein misfolding templated by pathogenic mutants
Many neurodegenerative diseases feature misfolded proteins that propagate via templated conversion of natively folded molecules. However, crucial questions about how such prion-like conversion occurs and what drives it remain unsolved, partly because technical challenges have prevented direct observation of conversion for any protein. We observed prion-like conversion in single molecules of superoxide dismutase-1 (SOD1), whose misfolding is linked to amyotrophic lateral sclerosis. Tethering pathogenic misfolded SOD1 mutants to wild-type molecules held in optical tweezers, we found that the mutants vastly increased misfolding of the wild-type molecule, inducing multiple misfolded isoforms. Crucially, the pattern of misfolding was the same in the mutant and converted wild-type domains and varied when the misfolded mutant was changed, reflecting the templating effect expected for prion-like conversion. Ensemble measurements showed decreased enzymatic activity in tethered heterodimers as conversion progressed, mirroring the single-molecule results. Antibodies sensitive to disease-specific epitopes bound to the converted protein, implying that conversion produced disease-relevant misfolded conformers. Protein misfolding can spread from one molecule to another in infectious prion diseases. The propagation of protein misfolding has been directly observed in single protein molecules. These results showed that pathogenic mutants of the protein superoxide dismutase-1 (SOD1), which causes familial amyotrophic lateral sclerosis, imprint their misfolding onto native wild-type molecules.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature chemical biology
生物-生化与分子生物学
CiteScore
23.90
自引率
1.40%
发文量
238
审稿时长
12 months
期刊介绍:
Nature Chemical Biology stands as an esteemed international monthly journal, offering a prominent platform for the chemical biology community to showcase top-tier original research and commentary. Operating at the crossroads of chemistry, biology, and related disciplines, chemical biology utilizes scientific ideas and approaches to comprehend and manipulate biological systems with molecular precision.
The journal embraces contributions from the growing community of chemical biologists, encompassing insights from chemists applying principles and tools to biological inquiries and biologists striving to comprehend and control molecular-level biological processes. We prioritize studies unveiling significant conceptual or practical advancements in areas where chemistry and biology intersect, emphasizing basic research, especially those reporting novel chemical or biological tools and offering profound molecular-level insights into underlying biological mechanisms.
Nature Chemical Biology also welcomes manuscripts describing applied molecular studies at the chemistry-biology interface due to the broad utility of chemical biology approaches in manipulating or engineering biological systems. Irrespective of scientific focus, we actively seek submissions that creatively blend chemistry and biology, particularly those providing substantial conceptual or methodological breakthroughs with the potential to open innovative research avenues. The journal maintains a robust and impartial review process, emphasizing thorough chemical and biological characterization.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


