吲哚 + NO3 有机气溶胶中硝基吲哚发色团的异构体鉴定
IF 3.7
Q2 CHEMISTRY, PHYSICAL
引用次数: 0
摘要
之前有人提出,硝酸根(NO3)氧化吲哚会生成硝基吲哚,而硝基吲哚是造成吲哚二次有机气溶胶(SOA)呈现棕色的主要原因。由于已知的硝基吲哚异构体有七种,我们使用色谱分离法证明在吲哚+NO3 反应中生成了一种单一的硝基吲哚异构体,并通过与硝基吲哚标准物质的色谱图进行比较,最终将其归类为 3-硝基吲哚。利用气溶胶质谱仪记录了气溶胶中 3-硝基吲哚颗粒的质谱,并将其与烟雾室中 NO3 氧化吲哚产生的 SOA 的质谱直接进行比较,以帮助确定硝基吲哚的独特峰值(m/z 162、132 和 116)。还进行了量子化学计算,以确定假设的吲哚 + NO3 中间体和产物的能量。这些数据的综合表明了一种机制,即首先从吲哚中的 N-H 键抽取一个氢原子,然后异构化为 3 位的碳中心自由基,接着加入 NO2。据预测,涉及从 C-H 键直接抽取一个氢原子或环的 NO3 加成的其他机制在能量上是不利的,因为初始反应步骤的障碍较大。本文章由计算机程序翻译,如有差异,请以英文原文为准。
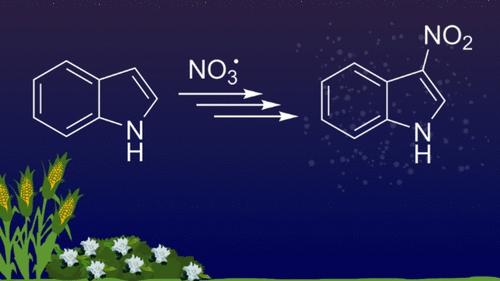
Isomeric Identification of the Nitroindole Chromophore in Indole + NO3 Organic Aerosol
Oxidation of indole by nitrate radical (NO3) was previously proposed to form nitroindole, largely responsible for the brown color of indole secondary organic aerosol (SOA). As there are seven known nitroindole isomers, we used chromatographic separation to show that a single nitroindole isomer is produced in the indole + NO3 reaction and definitively assigned it to 3-nitroindole by comparison with chromatograms of nitroindole standards. Mass spectra of aerosolized 3-nitroindole particles were recorded with an aerosol mass spectrometer and directly compared to mass spectra of SOA from smog chamber oxidation of indole by NO3 in order to help identify peaks unique to nitroindole (m/z 162, 132, and 116). Quantum chemical calculations were done to determine the energetics of hypothesized indole + NO3 intermediates and products. The combination of these data suggests a mechanism, wherein a hydrogen atom is first abstracted from the N–H bond in indole, followed by isomerization to a carbon-centered radical in the 3-position and followed by addition of NO2. Alternative mechanisms involving a direct abstraction of a H atom from a C–H bond or a NO3 addition to the ring are predicted to be energetically unfavorable from large barriers for the initial reaction steps.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
3.70
自引率
0.00%
发文量
0
期刊介绍:
ACS Physical Chemistry Au is an open access journal which publishes original fundamental and applied research on all aspects of physical chemistry. The journal publishes new and original experimental computational and theoretical research of interest to physical chemists biophysical chemists chemical physicists physicists material scientists and engineers. An essential criterion for acceptance is that the manuscript provides new physical insight or develops new tools and methods of general interest. Some major topical areas include:Molecules Clusters and Aerosols; Biophysics Biomaterials Liquids and Soft Matter; Energy Materials and Catalysis

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


