利用疏水性深共晶溶剂改进稀土元素的分离:从液液萃取到选择性溶解
IF 9.3
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
制备了由β-二酮和具有不同烷基链长的膦氧化物组成的深共晶溶剂(DES),并将其用作液-液萃取和溶解介质,用于分离铁、钴、钕和镝。在溶解过程中,立体受阻程度较高的 DES 对钕的选择性增强,而使用液液萃取法分离钕的效果较差。立体阻碍较小的 DES 的溶解选择性可通过添加不同烷基链长的有机酸来控制。本文章由计算机程序翻译,如有差异,请以英文原文为准。
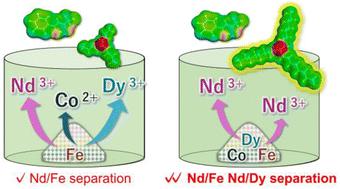
Improved separation of rare earth elements using hydrophobic deep eutectic solvents: liquid–liquid extraction to selective dissolution†
Deep eutectic solvents (DESs) composed of a beta-diketone and phosphine oxides featuring different alkyl chain lengths were prepared and then employed as liquid–liquid extraction and dissolution media for the separation of iron, cobalt, neodymium, and dysprosium. In the dissolution the more sterically hindered DESs exhibited enhanced selectivity to neodymium, which was poorly separated using liquid–liquid extraction. The dissolution selectivity in less sterically hindered DESs could be controlled by the addition of organic acids with different alkyl chain lengths.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Green Chemistry
化学-化学综合
CiteScore
16.10
自引率
7.10%
发文量
677
审稿时长
1.4 months
期刊介绍:
Green Chemistry is a journal that provides a unique forum for the publication of innovative research on the development of alternative green and sustainable technologies. The scope of Green Chemistry is based on the definition proposed by Anastas and Warner (Green Chemistry: Theory and Practice, P T Anastas and J C Warner, Oxford University Press, Oxford, 1998), which defines green chemistry as the utilisation of a set of principles that reduces or eliminates the use or generation of hazardous substances in the design, manufacture and application of chemical products. Green Chemistry aims to reduce the environmental impact of the chemical enterprise by developing a technology base that is inherently non-toxic to living things and the environment. The journal welcomes submissions on all aspects of research relating to this endeavor and publishes original and significant cutting-edge research that is likely to be of wide general appeal. For a work to be published, it must present a significant advance in green chemistry, including a comparison with existing methods and a demonstration of advantages over those methods.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


