噬菌体通过 RNA 和 DNA 结合的螺旋转螺旋蛋白进行抗CRISPR 控制。
IF 50.5
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
摘要
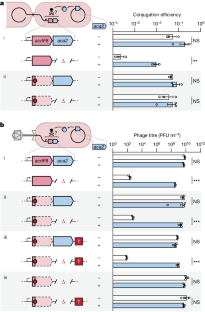
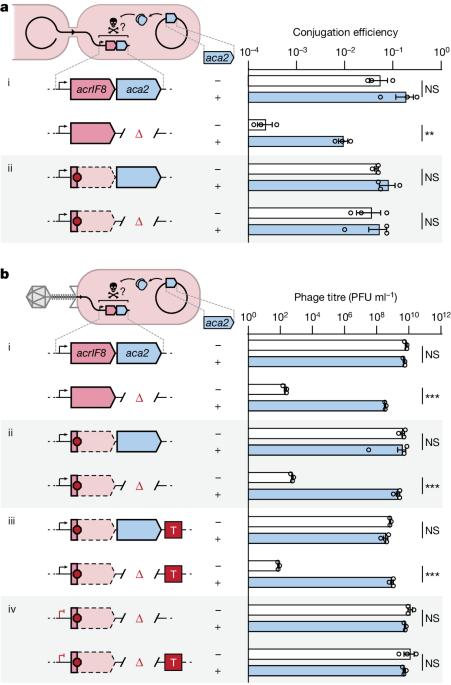
在所有生物体中,基因表达的调控都必须根据细胞的需要进行调整,这通常涉及到螺旋-翻转-螺旋(HTH)结构域蛋白1。例如,在细菌和噬菌体之间的军备竞赛中,噬菌体的抗CRISPR(acr)基因在感染后迅速表达,从而逃避CRISPR-Cas的防御;转录随后被含有HTH结构域的抗CRISPR相关(Aca)蛋白抑制,这可能是为了降低过度表达带来的健康代价2-5。然而,单个 HTH 调节器如何调整抗CRISPR 的产生以应对噬菌体基因组拷贝的增加和 acr mRNA 的积累,目前尚不清楚。在这里,我们发现调节因子 Aca2 的 HTH 结构域除了通过 DNA 结合抑制 Acr 的转录合成外,还通过结合保守的 RNA 干环和阻断核糖体的进入来抑制 mRNA 的翻译。约 40 kDa Aca2-RNA 复合物的冷冻电子显微镜结构展示了多功能 HTH 结构域如何从 DNA 结合位点特异性地识别 RNA。这些组合调控模式在 Aca2 家族中非常普遍,有助于在噬菌体 DNA 快速复制的情况下抑制 CRISPR-Cas,而不会出现有毒的 acr 过表达。鉴于含 HTH 域蛋白的普遍性,预计还会有更多的含 HTH 域蛋白通过 DNA 和 RNA 的双重结合进行调控。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Phage anti-CRISPR control by an RNA- and DNA-binding helix–turn–helix protein
In all organisms, regulation of gene expression must be adjusted to meet cellular requirements and frequently involves helix–turn–helix (HTH) domain proteins1. For instance, in the arms race between bacteria and bacteriophages, rapid expression of phage anti-CRISPR (acr) genes upon infection enables evasion from CRISPR–Cas defence; transcription is then repressed by an HTH-domain-containing anti-CRISPR-associated (Aca) protein, probably to reduce fitness costs from excessive expression2–5. However, how a single HTH regulator adjusts anti-CRISPR production to cope with increasing phage genome copies and accumulating acr mRNA is unknown. Here we show that the HTH domain of the regulator Aca2, in addition to repressing Acr synthesis transcriptionally through DNA binding, inhibits translation of mRNAs by binding conserved RNA stem-loops and blocking ribosome access. The cryo-electron microscopy structure of the approximately 40 kDa Aca2–RNA complex demonstrates how the versatile HTH domain specifically discriminates RNA from DNA binding sites. These combined regulatory modes are widespread in the Aca2 family and facilitate CRISPR–Cas inhibition in the face of rapid phage DNA replication without toxic acr overexpression. Given the ubiquity of HTH-domain-containing proteins, it is anticipated that many more of them elicit regulatory control by dual DNA and RNA binding. The helix–turn–helix domain of an anti-CRISPR-associated (Aca) protein represses transcription of anti-CRISPR (Acr)-encoding genes and inhibits their translation to protein via distinct binding modes to DNA and RNA, respectively.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature
综合性期刊-综合性期刊
CiteScore
90.00
自引率
1.20%
发文量
3652
审稿时长
3 months
期刊介绍:
Nature is a prestigious international journal that publishes peer-reviewed research in various scientific and technological fields. The selection of articles is based on criteria such as originality, importance, interdisciplinary relevance, timeliness, accessibility, elegance, and surprising conclusions. In addition to showcasing significant scientific advances, Nature delivers rapid, authoritative, insightful news, and interpretation of current and upcoming trends impacting science, scientists, and the broader public. The journal serves a dual purpose: firstly, to promptly share noteworthy scientific advances and foster discussions among scientists, and secondly, to ensure the swift dissemination of scientific results globally, emphasizing their significance for knowledge, culture, and daily life.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


