Mn(I)-Catalyzed Carbon-Skeleton Rearrangement of Tertiary Alcohol-Based Aldol Reaction with Aldehydes.
IF 3.3
2区 化学
Q1 CHEMISTRY, ORGANIC
The Journal of Organic Chemistry
Pub Date : 2024-08-02
Epub Date: 2024-07-11
DOI:10.1021/acs.joc.4c00835
引用次数: 0
摘要
研究人员开发了一种锰催化配体定向 Csp3-Csp2 偶联叔烯丙基醇与芳基醛的方法。该方法提供了一种通过基于碳骨架重排的醛醇反应获得 1,5-二叉戊-1-烯-3-酮的有效方法。本文章由计算机程序翻译,如有差异,请以英文原文为准。
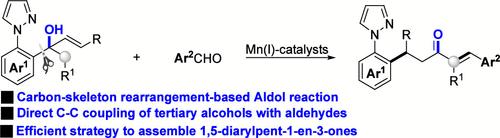
Mn(I)-Catalyzed Carbon-Skeleton Rearrangement of Tertiary Alcohol-Based Aldol Reaction with Aldehydes.
A Mn-catalyzed ligand-directed Csp3-Csp2 coupling of tertiary allylic alcohols with arylaldehydes has been developed. The method provides an efficient approach to access 1,5-diarylpent-1-en-3-ones via carbon-skeleton rearrangement-based aldol reaction.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

The Journal of Organic Chemistry
化学-有机化学
CiteScore
6.20
自引率
11.10%
发文量
1467
审稿时长
2 months
期刊介绍:
The Journal of Organic Chemistry welcomes original contributions of fundamental research in all branches of the theory and practice of organic chemistry. In selecting manuscripts for publication, the editors place emphasis on the quality and novelty of the work, as well as the breadth of interest to the organic chemistry community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


