利用内切酶 V 免疫染色测定 (EndoVIA) 观察细胞中 A 到 I 编辑的空间可视化情况
IF 12.7
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
腺苷-肌苷(A-to-I)编辑是最广泛的转录后 RNA 修饰之一,由作用于 RNA 的腺苷脱氨酶(ADARs)催化。不同组织类型的 A 到 I 编辑对多种生物功能至关重要,失调会导致自身免疫和神经系统疾病以及癌症。最近的证据还揭示了 RNA 定位与 A 到 I 编辑之间的联系,但人们对这种关系的内在机制及其生物学影响的了解仍然有限。目前的方法主要依赖于对提取的 RNA 进行体外表征,这最终会消除亚细胞定位和细胞间的异质性。为了应对这些挑战,我们重新利用了内切核酸酶 V(EndoV)--一种依赖于镁的核糖核酸酶,它能裂解编辑过的 RNA 中的肌苷酸碱基--来选择性地结合和检测细胞中 A 到 I 编辑过的 RNA。本文的研究工作介绍了一种内切酶 V 免疫染色测定(EndoVIA),该工作流程可提供编辑转录本的空间可视化,实现整体肌苷丰度的快速量化,并在纳米级水平上绘制转录本组中 A 到 I 编辑的全貌。本文章由计算机程序翻译,如有差异,请以英文原文为准。
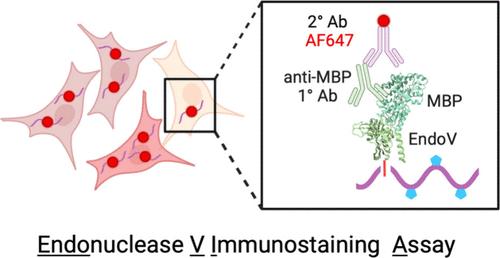
Spatial Visualization of A-to-I Editing in Cells Using Endonuclease V Immunostaining Assay (EndoVIA)
Adenosine-to-inosine (A-to-I) editing is one of the most widespread post-transcriptional RNA modifications and is catalyzed by adenosine deaminases acting on RNA (ADARs). Varying across tissue types, A-to-I editing is essential for numerous biological functions, and dysregulation leads to autoimmune and neurological disorders, as well as cancer. Recent evidence has also revealed a link between RNA localization and A-to-I editing, yet understanding of the mechanisms underlying this relationship and its biological impact remains limited. Current methods rely primarily on in vitro characterization of extracted RNA that ultimately erases subcellular localization and cell-to-cell heterogeneity. To address these challenges, we have repurposed endonuclease V (EndoV), a magnesium-dependent ribonuclease that cleaves inosine bases in edited RNA, to selectively bind and detect A-to-I edited RNA in cells. The work herein introduces an endonuclease V immunostaining assay (EndoVIA), a workflow that provides spatial visualization of edited transcripts, enables rapid quantification of overall inosine abundance, and maps the landscape of A-to-I editing within the transcriptome at the nanoscopic level.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

ACS Central Science
Chemical Engineering-General Chemical Engineering
CiteScore
25.50
自引率
0.50%
发文量
194
审稿时长
10 weeks
期刊介绍:
ACS Central Science publishes significant primary reports on research in chemistry and allied fields where chemical approaches are pivotal. As the first fully open-access journal by the American Chemical Society, it covers compelling and important contributions to the broad chemistry and scientific community. "Central science," a term popularized nearly 40 years ago, emphasizes chemistry's central role in connecting physical and life sciences, and fundamental sciences with applied disciplines like medicine and engineering. The journal focuses on exceptional quality articles, addressing advances in fundamental chemistry and interdisciplinary research.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


