多孔电极中 pH 缓冲氢气进化反应的两相质量输运动力学计算模型
IF 3.8
3区 工程技术
Q2 ENGINEERING, CHEMICAL
引用次数: 0
摘要
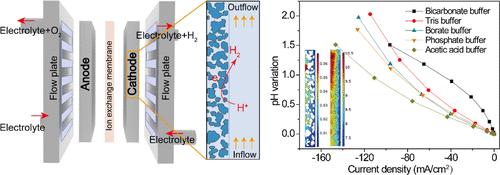
中性/近中性水电解有望降低工业制氢的总体成本。然而,目前的研究很少涉及多孔电极中氢气进化反应(HER)过程中化学/电化学反应与两相质量传输之间的耦合相互作用。在这项研究中,通过随机生长方法建立了多孔电极的几何模型,然后将数学模型纳入几何模型,系统地研究了这些不同过程之间的相互作用。对所建立的模型进行求解,以研究各种条件下的速度和浓度曲线。研究表明,与速度(电流密度增加 24%)相比,缓冲液浓度对 HER 性能的影响更为显著(电流密度增加 117%)。此外,由于离子和气泡的大量传输,流过式电极的 HER 电流密度比流过式电极高出 164%。此外,对比研究还表明,醋酸缓冲液中的电极具有最低的 pH 极化,从而最大限度地减少了 HER 过程中的质量传输损失,与其他缓冲液相比,HER 性能提高了 53%。这些结果表明,缓冲液物种和电极孔隙中气泡的质量传输可显著提高 HER 性能。本文的结论是,所建立的模型可以为多孔电极的设计和 HER 缓冲剂的选择提供一些指导,最终推进对多孔电极缓冲 HER 基本机制的理解。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Computational Model of Two-Phase Mass Transport Dynamics for pH-Buffered Hydrogen Evolution Reactions in Porous Electrodes
Neutral/near-neutral water electrolysis is promising to reduce the overall cost of industrial hydrogen production. However, the current studies rarely concern the coupled interactions among the chemical/electrochemical reactions and two-phase mass transport during the hydrogen evolution reaction (HER) in the porous electrode. In this work, the geometry of a porous electrode is developed through a random growth method, and the mathematical model is then incorporated into the geometrical model to systematically investigate the interplay among these diverse processes. The developed model is solved to examine velocity and concentration profiles under various conditions. The study shows that the buffer concentration has a much more remarkable effect (117% increase in current density) on the HER performance compared to velocity (24% increase in current density). In addition, the electrode with a flow-through mode delivers a 164% higher HER current density than that with flow-by mode due to the prompted mass transport of ions and bubbles. Furthermore, the comparison study suggests that the electrode in acetic acid buffer delivers the lowest pH polarization, thereby minimizing the mass transport loss during the HER and achieving a 53% improvement in HER performance compared to that in other buffers. These results indicate that the mass transport of buffer species and bubbles in the pores of the electrode can significantly improve the HER performance. This paper concludes that the developed model can provide some guidance for the design of a porous electrode and the selection of buffer for the HER, finally advancing the understanding of the fundamental mechanisms in the buffered HER of the porous electrode.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Industrial & Engineering Chemistry Research
工程技术-工程:化工
CiteScore
7.40
自引率
7.10%
发文量
1467
审稿时长
2.8 months
期刊介绍:
ndustrial & Engineering Chemistry, with variations in title and format, has been published since 1909 by the American Chemical Society. Industrial & Engineering Chemistry Research is a weekly publication that reports industrial and academic research in the broad fields of applied chemistry and chemical engineering with special focus on fundamentals, processes, and products.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


