富二氧化碳流中盐分、杂质和低含水量对天然气水合物形成的影响
IF 2
3区 工程技术
Q3 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
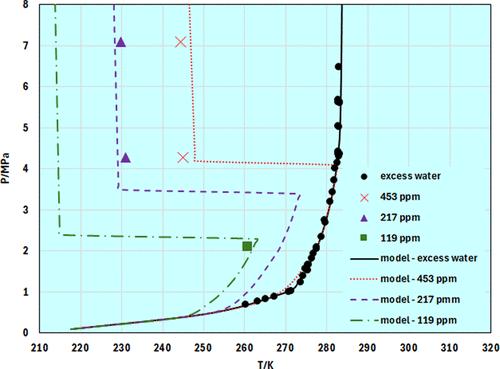
碳捕集利用和封存(CCUS)以及从高二氧化碳含量储层中生产天然气的做法,引起了人们对富含二氧化碳的天然气水合物形成的关注。这些气体混合物可能含有 Ar、O2、N2、H2、NxOy、CO、H2S、SO2 或汞等杂质,其中一些也是水合物形成物或潜在的腐蚀促进剂。本文探讨了如何利用热力学模型来预测富含二氧化碳的气流的相态。本文从二氧化碳与水的整体二元相平衡入手。然后观察气体水合物和不同杂质对水合物相行为的影响。此外,还分析了盐类对二氧化碳溶解度的影响以及盐类等抑制剂对二氧化碳水合物抑制的影响。在建立水合物模型时,使用了 Parrish 和 Prausnitz 实现的范德瓦耳斯和普拉蒂乌夫固溶体理论,并结合立方-加-关联状态方程(CPA EoS)来表示流体相。电解质对盐水中水合物形成和二氧化碳溶解度的影响是通过基于 Debye-Hückel 理论的附加电解质项来模拟的。该视角的主要目的是同时考虑所有可能的相平衡,因为在 CCUS 应用中,我们无法分离不同的相平衡条件。结果表明,所选模型可以准确地表示富二氧化碳流的相行为以及盐和杂质对气体水合物形成的影响。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Effect of Salts, Impurities, and Low Water Contents in the Formation of Gas Hydrates in CO2-Rich Streams
Carbon capture utilization and storage (CCUS) together with natural gas production from high CO2-containing reservoirs has raised attention to the formation of CO2-rich gas hydrates. These gas mixtures can contain impurities such as Ar, O2, N2, H2, NxOy, CO, H2S, SO2, or mercury, some of which are also hydrate formers or potential corrosion enhancers. This paper addresses the use of thermodynamic models to predict the phase behavior of CO2-rich streams. It starts from the overall binary phase equilibria of CO2 with water. Then, gas hydrates and the effect of different impurities on the hydrate phase behavior are observed. Furthermore, the effect of salts on CO2 solubility and the influence of inhibitors such as salts on CO2 hydrate suppression are analyzed. For modeling hydrates, the solid solution theory of van der Waals and Platteeuw, as implemented by Parrish and Prausnitz, is used coupled with the Cubic-Plus-Association equation of state (CPA EoS) for representing the fluid phases. The influence of electrolytes on hydrate formation and CO2 solubility in brines is modeled with an add-on electrolyte term based on the Debye–Hückel theory. The main objective of this Perspective is to consider all possible phase equilibria simultaneously, as in CCUS applications we cannot detach the different phase equilibria conditions. The results demonstrate that the selected models can accurately represent the phase behavior of CO2-rich streams and the effect of salts and impurities on gas hydrate formation.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Chemical & Engineering Data
工程技术-工程:化工
CiteScore
5.20
自引率
19.20%
发文量
324
审稿时长
2.2 months
期刊介绍:
The Journal of Chemical & Engineering Data is a monthly journal devoted to the publication of data obtained from both experiment and computation, which are viewed as complementary. It is the only American Chemical Society journal primarily concerned with articles containing data on the phase behavior and the physical, thermodynamic, and transport properties of well-defined materials, including complex mixtures of known compositions. While environmental and biological samples are of interest, their compositions must be known and reproducible. As a result, adsorption on natural product materials does not generally fit within the scope of Journal of Chemical & Engineering Data.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


