不同温度下氟化钾在八种单一溶剂和四种二元溶剂混合物中的溶解度测定及相关性分析
IF 2
3区 工程技术
Q3 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
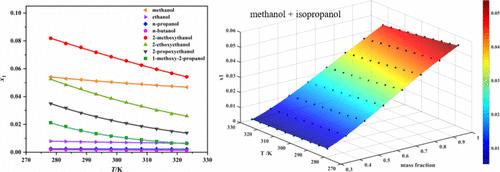
本研究采用重量法研究了氟化钾在八种单一溶剂(甲醇、乙醇、正丙醇、正丁醇、2-甲氧基乙醇、2-乙氧基乙醇、2-丙氧基乙醇和 1-甲氧基-2-丙醇)和四种二元溶剂混合物(甲醇 + 异丙醇、甲醇 + 丙酮、甲醇 + 乙腈和甲醇 + 乙酸乙酯)中的摩尔分数溶解度,温度范围为 278.15 至 323.15 K(间隔 5 K)。结果表明,在所有研究的溶剂体系中,氟化钾的溶解度随着温度的升高而逐渐降低。对于所研究的四种二元溶剂混合物,在温度不变的情况下,氟化钾的溶解度随着甲醇质量分数的增加而增加。实验溶解度数据与四个经验模型(van't Hoff 模型、改进的 Apelblat 模型、CNIBS/R-K 模型和 Jouyban-Acree-Apelblat 模型)相关联,所有模型都表现出优异的拟合性能。比较不同模型的拟合结果发现,改进的 Apelblat 模型的相对偏差(ARD)和均方根偏差(RMSD)值较低,表明其拟合精度较高,适合拟合选定溶剂中氟化钾的实验数据。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Determination and Correlation of the Solubility of Potassium Fluoride in Eight Single Solvents and Four Binary Solvent Mixtures at Different Temperatures
This study investigated the mole fraction solubility of potassium fluoride in eight single solvents (methanol, ethanol, n-propanol, n-butanol, 2-methoxyethanol, 2-ethoxyethanol, 2-propoxyethanol, and 1-methoxy-2-propanol) and four binary solvent mixtures (methanol + isopropanol, methanol + acetone, methanol + acetonitrile, and methanol + ethyl acetate) using the gravimetric method within the temperature range of 278.15 to 323.15 K (with 5 K intervals). The results demonstrated a gradual decrease in the solubility of potassium fluoride with increasing temperatures across all investigated solvent systems. For the four binary solvent mixtures studied, the solubility of potassium fluoride exhibited an increase with a higher mass fraction of methanol at a constant temperature. The experimental solubility data were correlated using four empirical models (van’t Hoff model, modified Apelblat model, CNIBS/R-K model, and Jouyban–Acree–Apelblat model), all of which exhibited excellent fitting performance. Comparison of different model fitting results revealed that the modified Apelblat model yielded lower values for relative deviation (ARD) and root-mean-square deviation (RMSD), indicating its superior fitting accuracy and suitability for fitting the experimental data of potassium fluoride in the selected solvents.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Chemical & Engineering Data
工程技术-工程:化工
CiteScore
5.20
自引率
19.20%
发文量
324
审稿时长
2.2 months
期刊介绍:
The Journal of Chemical & Engineering Data is a monthly journal devoted to the publication of data obtained from both experiment and computation, which are viewed as complementary. It is the only American Chemical Society journal primarily concerned with articles containing data on the phase behavior and the physical, thermodynamic, and transport properties of well-defined materials, including complex mixtures of known compositions. While environmental and biological samples are of interest, their compositions must be known and reproducible. As a result, adsorption on natural product materials does not generally fit within the scope of Journal of Chemical & Engineering Data.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


