发现类似于细胞周期蛋白依赖性激酶 2 的化学探针并确定其特性
IF 4
3区 医学
Q2 CHEMISTRY, MEDICINAL
引用次数: 0
摘要
通过分析无细胞结合和选择性数据,确定了基于酰氨基吲唑的 CDKL2 抑制剂。化合物 9 被选为 CDKL2 化学探针,因为它能有效抑制 CDKL2 的酶活性,与细胞中的 CDKL2 结合,并具有出色的全激酶组选择性,尤其是在细胞中使用时。化合物 16 被设计为阴性对照,在研究 CDKL2 介导的生物学实验中与化合物 9 同时使用。化合物 9 与 CDKL2 结合后的共晶体结构突显了其 ATP 结合位点内的关键相互作用。在大鼠原代神经元中抑制 CDKL2 底物 EB2 的下游磷酸化,证明化合物 9 在细胞中与 CDKL2 结合会抑制其活性。以相关浓度使用时,化合物 9 不会影响大鼠原代神经元或某些乳腺癌细胞的存活能力,也不会引起参与上皮-间质转化的蛋白质表达发生一致的变化。本文章由计算机程序翻译,如有差异,请以英文原文为准。
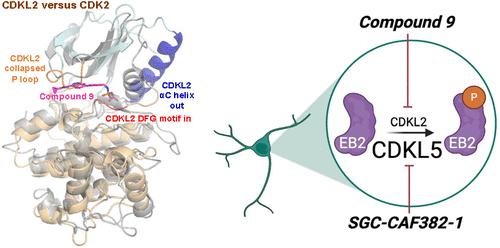
Discovery and Characterization of a Chemical Probe for Cyclin-Dependent Kinase-Like 2
Acylaminoindazole-based inhibitors of CDKL2 were identified via analyses of cell-free binding and selectivity data. Compound 9 was selected as a CDKL2 chemical probe based on its potent inhibition of CDKL2 enzymatic activity, engagement of CDKL2 in cells, and excellent kinome-wide selectivity, especially when used in cells. Compound 16 was designed as a negative control to be used alongside compound 9 in experiments to interrogate CDKL2-mediated biology. A solved cocrystal structure of compound 9 bound to CDKL2 highlighted key interactions it makes within its ATP-binding site. Inhibition of downstream phosphorylation of EB2, a CDKL2 substrate, in rat primary neurons provided evidence that engagement of CDKL2 by compound 9 in cells resulted in inhibition of its activity. When used at relevant concentrations, compound 9 does not impact the viability of rat primary neurons or certain breast cancer cells nor elicit consistent changes in the expression of proteins involved in epithelial–mesenchymal transition.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
ACS Medicinal Chemistry Letters
CHEMISTRY, MEDICINAL-
CiteScore
7.30
自引率
2.40%
发文量
328
审稿时长
1 months
期刊介绍:
ACS Medicinal Chemistry Letters is interested in receiving manuscripts that discuss various aspects of medicinal chemistry. The journal will publish studies that pertain to a broad range of subject matter, including compound design and optimization, biological evaluation, drug delivery, imaging agents, and pharmacology of both small and large bioactive molecules. Specific areas include but are not limited to:
Identification, synthesis, and optimization of lead biologically active molecules and drugs (small molecules and biologics)
Biological characterization of new molecular entities in the context of drug discovery
Computational, cheminformatics, and structural studies for the identification or SAR analysis of bioactive molecules, ligands and their targets, etc.
Novel and improved methodologies, including radiation biochemistry, with broad application to medicinal chemistry
Discovery technologies for biologically active molecules from both synthetic and natural (plant and other) sources
Pharmacokinetic/pharmacodynamic studies that address mechanisms underlying drug disposition and response
Pharmacogenetic and pharmacogenomic studies used to enhance drug design and the translation of medicinal chemistry into the clinic
Mechanistic drug metabolism and regulation of metabolic enzyme gene expression
Chemistry patents relevant to the medicinal chemistry field.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


