环甲基化 N∧C∧N 铂(II)和钯(II)基三重发射极的分子刚性化
IF 2.5
3区 化学
Q2 CHEMISTRY, INORGANIC & NUCLEAR
引用次数: 0
摘要
为了开发具有高效磷光的过渡金属配合物,我们合成了环甲基化配合物[M(N∧C∧N)Cl](M = Pd 或 Pt),其吡啶基环的两个正交位置上带有胺基 NH(C6H5)(HLNHPh)或 NH(C6H5CH2)(HLNHBn),可与 Cl-配体形成分子内氢键。单晶 X 射线衍射仪证实了氢键的存在,并显示出金属中心周围扭曲的方形平面几何形状,由于 Cl- 配位体明显偏离配位平面,τ4 值约为 0.25。与钯相比,铂的长波紫外可见吸收能量增加。铂(II)配合物在 298 K(λmax = 520 nm)和 77 K 的溶液中是强烈的三重发射体。钯(II)衍生物在 77 K 的冷冻玻璃基质中显示出与铂配合物相似的振动结构磷光色带,但在 298 K 的液态溶液中却不发光。由于三叉 N∧C∧N 配体的笨重正交 RNH 基团、Cl- 配体以及 N-H-Cl-H-N 氢键的立体排斥作用,这两种结构都发生了扭曲。本文章由计算机程序翻译,如有差异,请以英文原文为准。
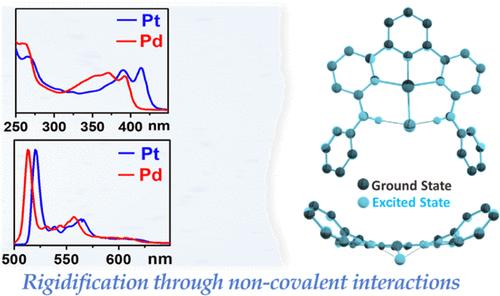
Molecular Rigidification of Cyclometalated N∧C∧N Pt(II)- and Pd(II)-Based Triplet Emitters
In an effort to develop transition metal complexes with efficient phosphorescence, we synthesized cyclometalated complexes [M(N∧C∧N)Cl] (M = Pd or Pt) with amine groups NH(C6H5) at the two ortho-positions of the pyridyl rings (HLNHPh) or NH(C6H5CH2) (HLNHBn) for intramolecular hydrogen bonding with the Cl– coligand. Single-crystal X-ray diffractometry confirmed the hydrogen bonding and showed a distorted square planar geometry around the metal centers with τ4 values of around 0.25 due to marked distortion of the Cl– coligand from the coordination plane. Long-wavelength UV–vis absorption energies are increased for Pt vs Pd. The Pt(II) complexes are intense triplet emitters in solution at 298 K (λmax = 520 nm) and at 77 K. The Pd(II) derivatives showed vibrationally structured phosphorescence bands in a frozen glassy matrix at 77 K resembling those of the Pt complexes but were nonemissive in liquid solutions at 298 K. The DFT-optimized electronic structures of the complexes show comparable geometries in their singlet ground state (S0) and in the first triplet (T1) excited states. Both are distorted by a combination of steric repulsion of the bulky ortho-RNH groups of the tridentate N∧C∧N ligands, the Cl– coligand, and the N–H···Cl···H–N hydrogen bonds.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Organometallics
化学-无机化学与核化学
CiteScore
5.60
自引率
7.10%
发文量
382
审稿时长
1.7 months
期刊介绍:
Organometallics is the flagship journal of organometallic chemistry and records progress in one of the most active fields of science, bridging organic and inorganic chemistry. The journal publishes Articles, Communications, Reviews, and Tutorials (instructional overviews) that depict research on the synthesis, structure, bonding, chemical reactivity, and reaction mechanisms for a variety of applications, including catalyst design and catalytic processes; main-group, transition-metal, and lanthanide and actinide metal chemistry; synthetic aspects of polymer science and materials science; and bioorganometallic chemistry.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


