在干眼发育过程中,昼夜节律紊乱会通过时钟分子 BMAL1 减少 MUC4 的表达。
IF 9.5
2区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
由于轮班工作、时差和其他生活方式因素造成的昼夜节律紊乱是一个常见的公共健康问题,与代谢紊乱、神经退行性疾病和癌症等多种疾病相关。在本研究中,我们使用每 3 天一次的时间转换方法建立了一个慢性时差模型,并评估了昼夜节律紊乱对眼表平衡的影响。结果表明,时差增加了角膜上皮缺陷、细胞凋亡和促炎细胞因子的表达。然而,时差 30 天后,泪液分泌量和结膜上皮细胞的数量并无明显变化。此外,利用 RNA 测序对致病机制的进一步分析表明,时差导致角膜跨膜粘蛋白缺乏,特别是 MUC4 缺乏。补充 MUC4 后,角膜上皮细胞得到保护,炎症激活也受到抑制,这证明了 MUC4 在致病过程中的关键作用。意想不到的是,小鼠BMAL1基因消减会导致MUC4缺乏和干眼症。在体外培养的人类角膜上皮细胞中,BMAL1沉默会减少MUC4的表达,而BMAL1过表达则会增加MUC4的表达。此外,褪黑激素是一种昼夜节律恢复剂,它能恢复 BMAL1 的表达,从而上调 MUC4,对时差引起的干眼症有治疗作用。因此,我们建立了一种由昼夜节律紊乱诱发的新型干眼症小鼠模型,阐明了其潜在机制,并确定了一种潜在的临床治疗方法。本文章由计算机程序翻译,如有差异,请以英文原文为准。
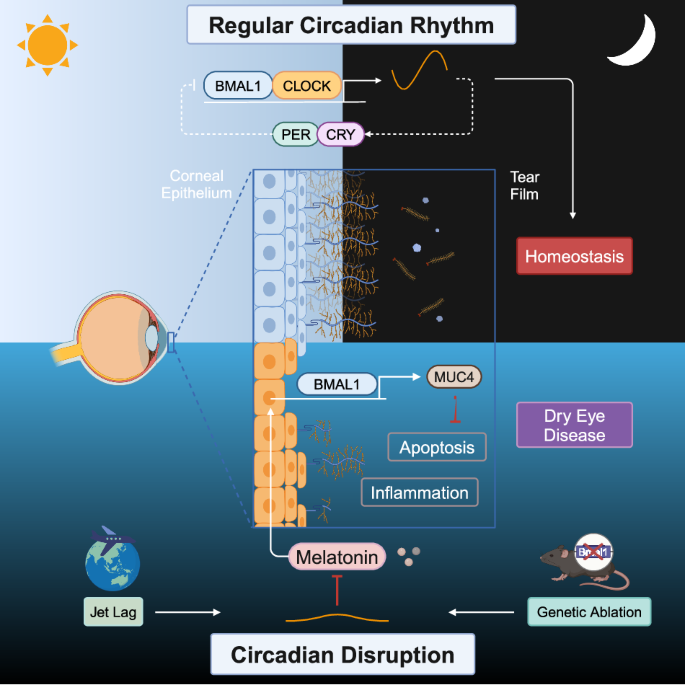
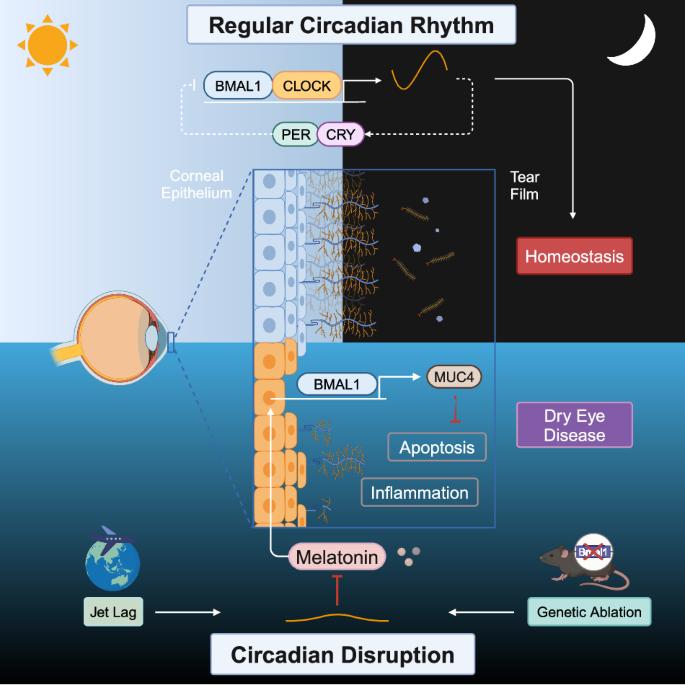
Circadian disruption reduces MUC4 expression via the clock molecule BMAL1 during dry eye development
Circadian disruption, as a result of shiftwork, jet lag, and other lifestyle factors, is a common public health problem associated with a wide range of diseases, such as metabolic disorders, neurodegenerative diseases, and cancer. In the present study, we established a chronic jet lag model using a time shift method every 3 days and assessed the effects of circadian disruption on ocular surface homeostasis. Our results indicated that jet lag increased corneal epithelial defects, cell apoptosis, and proinflammatory cytokine expression. However, the volume of tear secretion and the number of conjunctival goblet cells did not significantly change after 30 days of jet lag. Moreover, further analysis of the pathogenic mechanism using RNA sequencing revealed that jet lag caused corneal transmembrane mucin deficiency, specifically MUC4 deficiency. The crucial role of MUC4 in pathogenic progression was demonstrated by the protection of corneal epithelial cells and the inhibition of inflammatory activation following MUC4 replenishment. Unexpectedly, genetic ablation of BMAL1 in mice caused MUC4 deficiency and dry eye disease. The underlying mechanism was revealed in cultured human corneal epithelial cells in vitro, where BMAL1 silencing reduced MUC4 expression, and BMAL1 overexpression increased MUC4 expression. Furthermore, melatonin, a circadian rhythm restorer, had a therapeutic effect on jet lag-induced dry eye by restoring the expression of BMAL1, which upregulated MUC4. Thus, we generated a novel dry eye mouse model induced by circadian disruption, elucidated the underlying mechanism, and identified a potential clinical treatment. Dry eye disease, a long-term issue causing discomfort and vision problems, impacts millions globally. In this research, scientists studied how disturbances in our internal clock contribute to DED. Researchers made the mice experience an 8-hour shift in their day-night cycle every 3 days, imitating chronic jet lag. The findings showed that chronic jet lag resulted in a significant decrease in MUC4 expression in the cornea, leading to DED symptoms. Supplementing with MUC4 or treating the mice with melatonin, eased these symptoms. This indicates that disruptions to our internal clock can directly affect eye health by impacting key protective proteins in the eye. Researchers conclude that maintaining a healthy internal clock is vital for eye health and that treatments targeting internal clock disruptions could help DED patients. This summary was initially drafted using artificial intelligence, then revised and fact-checked by the author.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Experimental and Molecular Medicine
医学-生化与分子生物学
CiteScore
19.50
自引率
0.80%
发文量
166
审稿时长
3 months
期刊介绍:
Experimental & Molecular Medicine (EMM) stands as Korea's pioneering biochemistry journal, established in 1964 and rejuvenated in 1996 as an Open Access, fully peer-reviewed international journal. Dedicated to advancing translational research and showcasing recent breakthroughs in the biomedical realm, EMM invites submissions encompassing genetic, molecular, and cellular studies of human physiology and diseases. Emphasizing the correlation between experimental and translational research and enhanced clinical benefits, the journal actively encourages contributions employing specific molecular tools. Welcoming studies that bridge basic discoveries with clinical relevance, alongside articles demonstrating clear in vivo significance and novelty, Experimental & Molecular Medicine proudly serves as an open-access, online-only repository of cutting-edge medical research.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


