在铜催化的烯烃氢功能化过程中模拟氢原子转移反应
IF 42.8
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
摘要
催化金属氢化物氢原子转移(MHAT)的复兴为简单烯烃的自由基化学提供了先进的工具。虽然钴、铁和锰等三维过渡金属在催化氢原子转移方面已得到广泛研究,但铜的潜力仍未得到开发。这是因为经典的亲核 Cu(I)-H 具有极性反应。在此,我们报告了铜催化的类似 MHAT 的氧化加氢功能化反应。与传统的 Cu(I)-H 化学反应不同,推定的 Cu-MHAT 过程产生的烷基自由基具有很高的化学选择性和区域选择性,这些自由基随后被 Cu(II) 物种捕获,与多种氧、氮、卤素和碳基亲核物质发生偶联反应。初步研究结果表明,将其扩展到不对称催化和自由基聚合反应是可行的。这项工作提供了一个互补的氧化 MHAT 平台。本文章由计算机程序翻译,如有差异,请以英文原文为准。
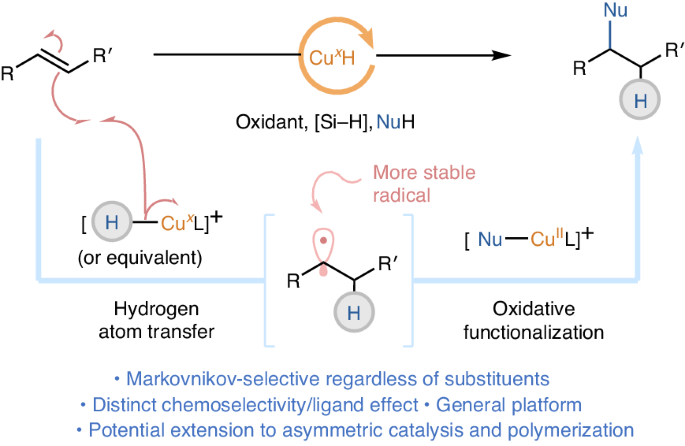
Mimicking hydrogen-atom-transfer-like reactivity in copper-catalysed olefin hydrofunctionalization
The renaissance of catalytic metal hydride hydrogen atom transfer (MHAT) offers advanced tools for radical chemistry on simple olefins. While 3d transition metals like cobalt, iron and manganese have been extensively studied in catalytic MHAT, the potential of copper remains unexplored. This is due to the polar reactivity exhibited by classical nucleophilic Cu(I)–H. Here we report copper-catalysed MHAT-like oxidative hydrofunctionalization reactions. In contrast to conventional Cu(I)–H chemistry, the putative Cu-MHAT process produces alkyl radicals with high chemoselectivity and regioselectivity, which are subsequently captured by Cu(II) species to undergo coupling reactions with a broad scope of oxygen-, nitrogen-, halogen- and carbon-based nucleophiles. Preliminary results suggest viable extension to asymmetric catalysis and radical polymerization. This work offers a complementary oxidative MHAT platform. Cobalt, iron or manganese catalysts are the metals of choice in alkene functionalization reactions via catalytic metal hydride hydrogen atom transfer (MHAT). Now a complementary MHAT-like system based on copper is proposed to operate in a regioselective Markovnikov addition reaction of nucleophiles to olefins.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature Catalysis
Chemical Engineering-Bioengineering
CiteScore
52.10
自引率
1.10%
发文量
140
期刊介绍:
Nature Catalysis serves as a platform for researchers across chemistry and related fields, focusing on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, encompassing both fundamental and applied studies. With a particular emphasis on advancing sustainable industries and processes, the journal provides comprehensive coverage of catalysis research, appealing to scientists, engineers, and researchers in academia and industry.
Maintaining the high standards of the Nature brand, Nature Catalysis boasts a dedicated team of professional editors, rigorous peer-review processes, and swift publication times, ensuring editorial independence and quality. The journal publishes work spanning heterogeneous catalysis, homogeneous catalysis, and biocatalysis, covering areas such as catalytic synthesis, mechanisms, characterization, computational studies, nanoparticle catalysis, electrocatalysis, photocatalysis, environmental catalysis, asymmetric catalysis, and various forms of organocatalysis.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


