银(I)催化和 DBU 促进丙炔醇与胺的异构化/加成,以获得 β-氨基酮
IF 1.5
4区 化学
Q3 CHEMISTRY, ORGANIC
引用次数: 0
摘要
通过银(I)催化和 DBU 促进的丙炔醇与胺的异构化/加成,实现了合成 β-氨基酮的另一种策略。其机理可能是异构化和顺序加成与烯基自由基过程相结合。该方法具有底物范围广、原子经济性好、操作简便、产率高至优等特点,为克级规模合成药物普罗罗生提供了一种新方法,为β-氨基酮的构建提供了实际应用。本文章由计算机程序翻译,如有差异,请以英文原文为准。
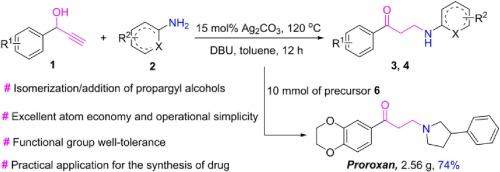
Silver(I)-catalyzed and DBU-promoted isomerization/addition of propargyl alcohols to amines to access β-aminoketones
An alternative strategy for the synthesis of β-aminoketones have been achieved through silver(I)-catalyzed and DBU-promoted isomerization/addition of propargyl alcohols to amines. The mechanism likely involves an isomerization and sequential addition combined with the alkenyl radical process. This protocol features broad substrate scope, superior atom economy, operational simplicity, and good to excellent yields, and provides a new method for the synthesis of drug Proroxan on gram-scale, presenting a practical application for the construction of β-aminoketones.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Tetrahedron Letters
化学-有机化学
CiteScore
3.50
自引率
5.60%
发文量
521
审稿时长
28 days
期刊介绍:
Tetrahedron Letters provides maximum dissemination of outstanding developments in organic chemistry. The journal is published weekly and covers developments in techniques, structures, methods and conclusions in experimental and theoretical organic chemistry. Rapid publication of timely and significant research results enables researchers from all over the world to transmit quickly their new contributions to large, international audiences.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


