来自互补配对炔烃的游离碳烯
IF 19.2
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
烯属化合物(R1R2C:)与自由基、芳香族化合物和亚硝基烯属化合物一样,是一个重要的中性、高能、反应性中间体家族--可进行快速反应的化学实体。炔(R3C≡CR4)是一种基本官能团,具有很高的势能;不过,炔的动力学稳定性很高,因此可以作为货架上稳定的化学商品方便地处理。如果能利用炔烃的高势能(即热力学势能)直接生成无金属的碳烯,将是一项巨大的进步。我们在此报告,只需加热 2-炔基亚氨基杂环(一种含有亲核氮原子的环状化合物)与亲电炔烃的混合物,就能实现这一目标。我们证明了这一过程具有相当大的通用性:许多货架上稳定的炔烃亲电体与许多种类的(2-炔基)杂环亲核体发生反应,生成碳烯中间体,这些中间体立即进行多种类型的转化,从而方便、实用地获得各种杂环产品。对这些反应的关键机理进行了阐述。本文章由计算机程序翻译,如有差异,请以英文原文为准。
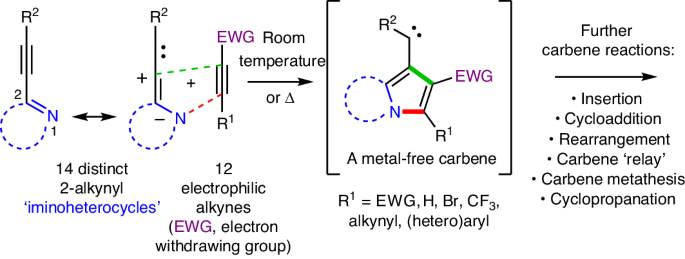
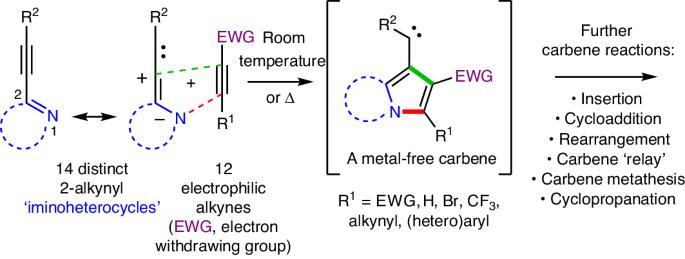
Free carbenes from complementarily paired alkynes
Carbenes (R1R2C:) like radicals, arynes and nitrenes constitute an important family of neutral, high-energy, reactive intermediates—fleeting chemical entities that undergo rapid reactions. An alkyne (R3C≡CR4) is a fundamental functional group that houses a high degree of potential energy; however, the substantial kinetic stability of alkynes renders them conveniently handleable as shelf-stable chemical commodities. The ability to generate metal-free carbenes directly from alkynes, fuelled by the high potential (that is, thermodynamic) energy of the latter, would constitute a considerable advance. We report here that this can be achieved simply by warming a mixture of a 2-alkynyl iminoheterocycle (a cyclic compound containing a nucleophilic nitrogen atom) with an electrophilic alkyne. We demonstrate considerable generality for the process: many shelf-stable alkyne electrophiles engage many classes of (2-alkynyl)heterocyclic nucleophiles to produce carbene intermediates that immediately undergo many types of transformations to provide facile and practical access to a diverse array of heterocyclic products. Key mechanistic aspects of the reactions are delineated. Thermal generation of free carbenes with 100% atom economy is rare. Now, it has been shown that this can be achieved by the net [3+2] cycloaddition between many classes of 2-alkynyl iminoheterocycles and electrophilic alkynes. Together with a host of carbene trapping reactions, this method provides facile and versatile access to diverse heterocycle scaffolds.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature chemistry
化学-化学综合
CiteScore
29.60
自引率
1.40%
发文量
226
审稿时长
1.7 months
期刊介绍:
Nature Chemistry is a monthly journal that publishes groundbreaking and significant research in all areas of chemistry. It covers traditional subjects such as analytical, inorganic, organic, and physical chemistry, as well as a wide range of other topics including catalysis, computational and theoretical chemistry, and environmental chemistry.
The journal also features interdisciplinary research at the interface of chemistry with biology, materials science, nanotechnology, and physics. Manuscripts detailing such multidisciplinary work are encouraged, as long as the central theme pertains to chemistry.
Aside from primary research, Nature Chemistry publishes review articles, news and views, research highlights from other journals, commentaries, book reviews, correspondence, and analysis of the broader chemical landscape. It also addresses crucial issues related to education, funding, policy, intellectual property, and the societal impact of chemistry.
Nature Chemistry is dedicated to ensuring the highest standards of original research through a fair and rigorous review process. It offers authors maximum visibility for their papers, access to a broad readership, exceptional copy editing and production standards, rapid publication, and independence from academic societies and other vested interests.
Overall, Nature Chemistry aims to be the authoritative voice of the global chemical community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


