TBK1适配体NAP1和SINTBAD控制有丝分裂的启动和进展
IF 12.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
有丝分裂通过选择性地靶向降解受损线粒体来保护线粒体的整体健康。防止PTEN诱导的推定激酶1(PINK1)和E3泛素连接酶Parkin(PINK1/Parkin)依赖的线粒体吞噬和其他选择性自噬途径反应过度,同时确保一旦启动就能迅速进行的调控机制在很大程度上是难以捉摸的。在这里,我们证明了 TBK1(TANK 结合激酶 1)适配体 NAP1(NAK 相关蛋白 1)和 SINTBAD(类似于 NAP1 的 TBK1 适配体)如何通过与 OPTN 竞争 TBK1 来限制 OPTN(optineurin)驱动的有丝分裂的启动。相反,它们通过将 TBK1 募集到 NDP52 并稳定其与 FIP200 的相互作用,促进核点状蛋白 52(NDP52)驱动的有丝分裂的进展。值得注意的是,OPTN 是有丝分裂启动过程中 TBK1 的主要招募者,而 TBK1 则反过来促进 NDP52 介导的有丝分裂。因此,我们的研究结果将 NAP1 和 SINTBAD 定义为货物受体流变器,可提高 OPTN 启动有丝分裂的阈值,同时在 NDP52 的支持下,一旦启动,就会促进该途径的进展。这些发现揭示了在确保高效进展的同时防止通路过度活跃的细胞策略。本文章由计算机程序翻译,如有差异,请以英文原文为准。
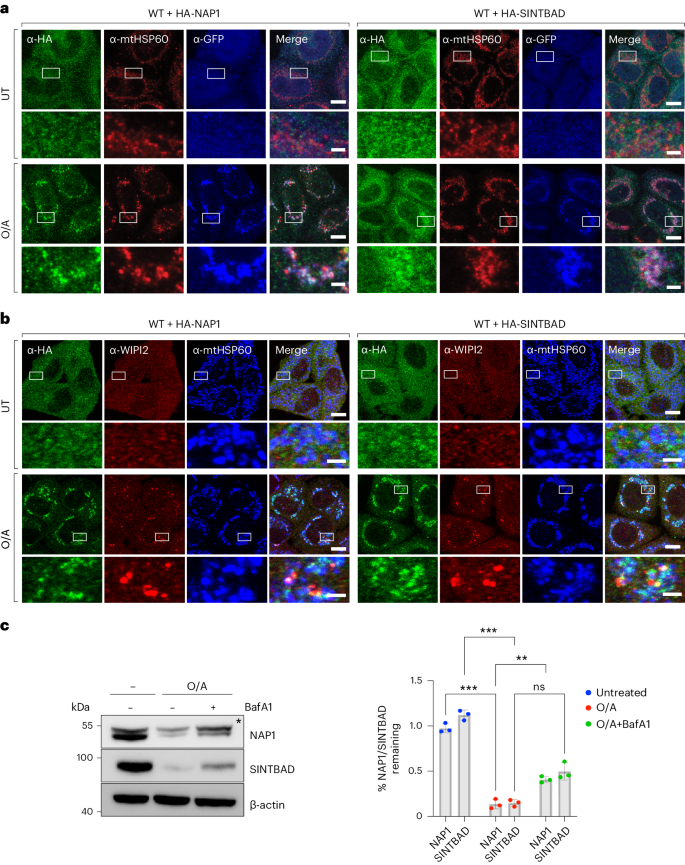
Control of mitophagy initiation and progression by the TBK1 adaptors NAP1 and SINTBAD
Mitophagy preserves overall mitochondrial fitness by selectively targeting damaged mitochondria for degradation. The regulatory mechanisms that prevent PTEN-induced putative kinase 1 (PINK1) and E3 ubiquitin ligase Parkin (PINK1/Parkin)-dependent mitophagy and other selective autophagy pathways from overreacting while ensuring swift progression once initiated are largely elusive. Here, we demonstrate how the TBK1 (TANK-binding kinase 1) adaptors NAP1 (NAK-associated protein 1) and SINTBAD (similar to NAP1 TBK1 adaptor) restrict the initiation of OPTN (optineurin)-driven mitophagy by competing with OPTN for TBK1. Conversely, they promote the progression of nuclear dot protein 52 (NDP52)-driven mitophagy by recruiting TBK1 to NDP52 and stabilizing its interaction with FIP200. Notably, OPTN emerges as the primary recruiter of TBK1 during mitophagy initiation, which in return boosts NDP52-mediated mitophagy. Our results thus define NAP1 and SINTBAD as cargo receptor rheostats, elevating the threshold for mitophagy initiation by OPTN while promoting the progression of the pathway once set in motion by supporting NDP52. These findings shed light on the cellular strategy to prevent pathway hyperactivity while still ensuring efficient progression. Mitophagy is an important quality control pathway. Here, the authors identify the mechanisms enabling the TBK1 adaptors NAP1 and SINTBAD to prevent hyperactivation of PINK1/Parkin mitophagy while promoting the pathway once set in motion.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature Structural & Molecular Biology
BIOCHEMISTRY & MOLECULAR BIOLOGY-BIOPHYSICS
CiteScore
22.00
自引率
1.80%
发文量
160
审稿时长
3-8 weeks
期刊介绍:
Nature Structural & Molecular Biology is a comprehensive platform that combines structural and molecular research. Our journal focuses on exploring the functional and mechanistic aspects of biological processes, emphasizing how molecular components collaborate to achieve a particular function. While structural data can shed light on these insights, our publication does not require them as a prerequisite.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


