用 CRISPR 毒素-抗毒素基因驱动技术超越拟南芥的孟德尔遗传,使花粉萌发受阻
IF 15.8
1区 生物学
Q1 PLANT SCIENCES
引用次数: 0
摘要
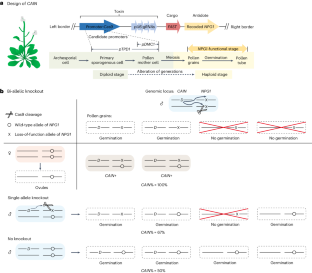
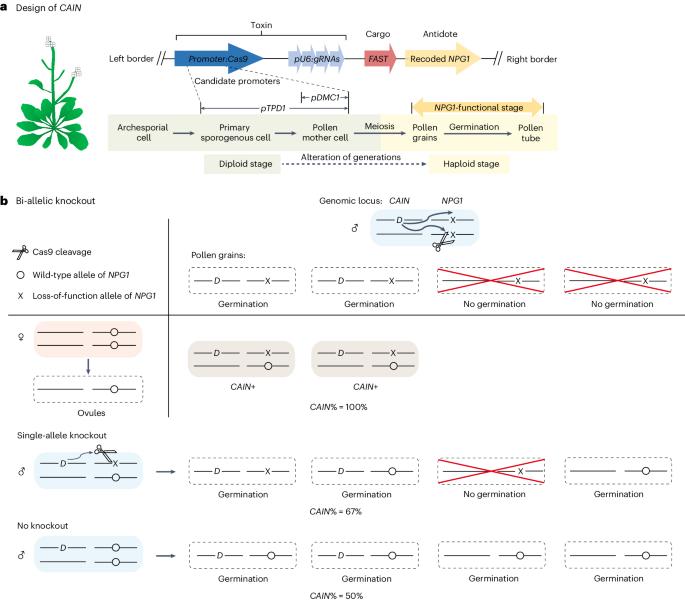
合成基因驱动受到自然自私遗传因子的启发,并以超孟德尔(50%)频率传递给后代,为在野生种群中传播有益于人类的性状提供了变革潜力,即使面临潜在的适应性成本。在这里,我们构建了一个名为 "利用 NPG1 的 CRISPR 辅助遗传"(CRISPR-Assisted Inheritance utilizing NPG1,CAIN)的植物基因驱动系统,该系统利用雄性种系中的毒素-解毒剂机制来推翻孟德尔遗传。具体来说,一个引导 RNA-Cas9 盒针对重要的无花粉萌发 1(NPG1)基因,作为阻止花粉萌发的毒素。一个重新编码的、抗 CRISPR 的 NPG1 拷贝可作为解毒剂,仅在携带驱动基因的花粉细胞中提供解救。为了限制无意释放可能造成的后果,我们使用了自花授粉的拟南芥作为模型。驱动力在连续两代中的传播率高达 88-99%,产生的抗性等位基因极少,不太可能抑制驱动力的传播。我们的研究为快速改变或抑制杂交植物种群的基因提供了坚实的基础。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Overriding Mendelian inheritance in Arabidopsis with a CRISPR toxin–antidote gene drive that impairs pollen germination
Synthetic gene drives, inspired by natural selfish genetic elements and transmitted to progeny at super-Mendelian (>50%) frequencies, present transformative potential for disseminating traits that benefit humans throughout wild populations, even facing potential fitness costs. Here we constructed a gene drive system in plants called CRISPR-Assisted Inheritance utilizing NPG1 (CAIN), which uses a toxin–antidote mechanism in the male germline to override Mendelian inheritance. Specifically, a guide RNA–Cas9 cassette targets the essential No Pollen Germination 1 (NPG1) gene, serving as the toxin to block pollen germination. A recoded, CRISPR-resistant copy of NPG1 serves as the antidote, providing rescue only in pollen cells that carry the drive. To limit potential consequences of inadvertent release, we used self-pollinating Arabidopsis thaliana as a model. The drive demonstrated a robust 88–99% transmission rate over two successive generations, producing minimal resistance alleles that are unlikely to inhibit drive spread. Our study provides a strong basis for rapid genetic modification or suppression of outcrossing plant populations. Researchers have developed a synthetic gene drive in Arabidopsis, demonstrating a proof of concept for biasing inheritance that could enable rapid genetic modifications or population suppression in wild plants.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
Nature Plants
PLANT SCIENCES-
CiteScore
25.30
自引率
2.20%
发文量
196
期刊介绍:
Nature Plants is an online-only, monthly journal publishing the best research on plants — from their evolution, development, metabolism and environmental interactions to their societal significance.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


