苯甲酰苯乙炔介导的吡啶与 H-膦酸盐的 SNHAr 无催化剂和无溶剂区域特异性膦酰化反应。
IF 2.9
3区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
摘要
在无催化剂和无溶剂条件下(50-55 °C),在苯甲酰苯乙炔的存在下,吡啶与 H-膦酸盐发生简便的 SNHAr 膦酰化反应,生成 4-膦酰基吡啶,收率高达 68%。在该反应中,苯甲酰苯乙炔通过形成 1,3(4)-二极络合物激活吡啶环,使 H-膦酸去质子化生成 P-中心阴离子,最后作为氧化剂从中间离子对中消除。在上述 SNHAr 过程中,末端缺电子乙炔(丙炔酸甲酯和苯甲酰乙炔)作为介质的效率很低。本文章由计算机程序翻译,如有差异,请以英文原文为准。
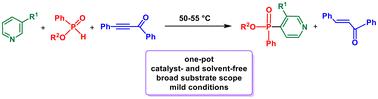
Catalyst- and solvent-free regiospecific SNHAr phosphinylation of pyridines with H-phosphinates mediated by benzoylphenylacetylene†
Pyridines undergo a facile SNHAr phosphinylation with H-phosphinates under catalyst- and solvent-free conditions (50–55 °C) in the presence of benzoylphenylacetylene to afford 4-phosphinylpyridines in up to 68% yield. In this reaction, benzoylphenylacetylene activates the pyridine ring by the formation of a 1,3(4)-dipolar complex, deprotonates H-phosphinates to generate P-centered anions and finally acts as an oxidizer, being eliminated from an intermediate ion pair. Terminal electron-deficient acetylenes (methyl propiolate and benzoylacetylene) are inefficient as mediators in the above SNHAr process.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Organic & Biomolecular Chemistry
化学-有机化学
CiteScore
5.50
自引率
9.40%
发文量
1056
审稿时长
1.3 months
期刊介绍:
The international home of synthetic, physical and biomolecular organic chemistry.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


