硅、铍和磷原子在 (101¯0) Re 表面化合物形成过程中的竞争
IF 2.1
4区 化学
Q3 CHEMISTRY, PHYSICAL
引用次数: 0
摘要
我们研究了三种不同吸附剂 Si、Be 和 P 在 (101¯0) Re 面上的高温联合吸附。在联合吸附过程中,其中一种吸附剂被转移到体液中,进入溶解状态。位移以原子到原子的方式发生,具有 "循环性质":硅位移到磷,铍位移到硅,磷位移到铍。本文章由计算机程序翻译,如有差异,请以英文原文为准。
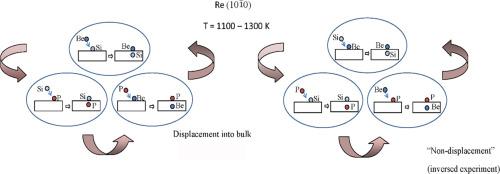
Competition between silicon, beryllium and phosphorus atoms in the formation of surface chemical compounds on (101¯0) Re
High-temperature joint adsorption has been studied of three different adsorbates, Si, Be, and P on the Re face. All three adsorbates form surface compounds with the stoichiometry ReX, where X is Si, Be, and P. In joint adsorption, one of the adsorbates is displaced into the bulk, into the dissolved state. The processes are observed at a temperature sufficient for diffusion in the bulk, 1100–1300 K. Displacement occurs in an atom-to-atom mode, and it has a “cyclic nature”: silicon displaces phosphorus, beryllium displaces silicon, and phosphorus - beryllium.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Surface Science
化学-物理:凝聚态物理
CiteScore
3.30
自引率
5.30%
发文量
137
审稿时长
25 days
期刊介绍:
Surface Science is devoted to elucidating the fundamental aspects of chemistry and physics occurring at a wide range of surfaces and interfaces and to disseminating this knowledge fast. The journal welcomes a broad spectrum of topics, including but not limited to:
• model systems (e.g. in Ultra High Vacuum) under well-controlled reactive conditions
• nanoscale science and engineering, including manipulation of matter at the atomic/molecular scale and assembly phenomena
• reactivity of surfaces as related to various applied areas including heterogeneous catalysis, chemistry at electrified interfaces, and semiconductors functionalization
• phenomena at interfaces relevant to energy storage and conversion, and fuels production and utilization
• surface reactivity for environmental protection and pollution remediation
• interactions at surfaces of soft matter, including polymers and biomaterials.
Both experimental and theoretical work, including modeling, is within the scope of the journal. Work published in Surface Science reaches a wide readership, from chemistry and physics to biology and materials science and engineering, providing an excellent forum for cross-fertilization of ideas and broad dissemination of scientific discoveries.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


