在抑制 mTORC1 的过程中,eIF4A1 可增强 LARP1 介导的翻译抑制作用
IF 12.5
1区 生物学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
真核生物翻译起始因子(eIF)4A--一种 DEAD-box RNA 结合蛋白--在翻译起始过程中发挥着至关重要的作用。最近的报道提出了 eIF4A 依赖螺旋酶和不依赖螺旋酶的功能,但 eIF4A 的多方面作用尚未得到充分探讨。在这里,我们发现 eIF4A1 在雷帕霉素机理靶点复合体 1(mTORC1)(一种控制细胞增殖的重要激酶复合体)受到抑制时会增强翻译抑制。RNA pulldown测序显示,eIF4A1优先与含有末端寡嘧啶(TOP)基团的mRNA结合,这些mRNA的翻译在mTORC1受到抑制时被迅速抑制。这种选择性相互作用依赖于一种与 La 相关的 RNA 结合蛋白 LARP1。核糖体分析表明,缺失 EIF4A1 会减弱 mTORC1 失活时对 TOP mRNA 的翻译抑制。此外,eIF4A1 增加了 TOP mRNA 与 LARP1 之间的相互作用,从而确保了 mTORC1 受抑制时更强的翻译抑制作用。我们的数据显示了 eIF4A1 通过抑制性结合伙伴调节蛋白质合成的多模式性,并为通用翻译激活因子的抑制作用提供了一个独特的例子。本文章由计算机程序翻译,如有差异,请以英文原文为准。
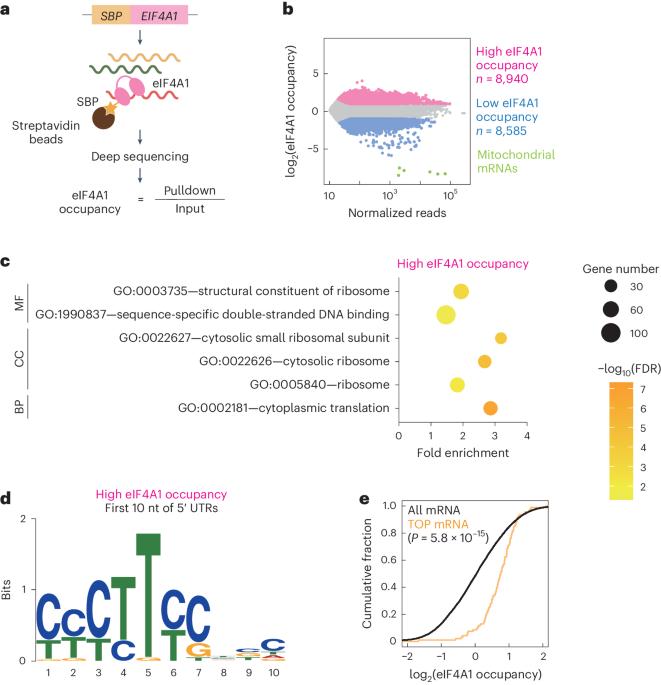
eIF4A1 enhances LARP1-mediated translational repression during mTORC1 inhibition
Eukaryotic translation initiation factor (eIF)4A—a DEAD-box RNA-binding protein—plays an essential role in translation initiation. Recent reports have suggested helicase-dependent and helicase-independent functions for eIF4A, but the multifaceted roles of eIF4A have not been fully explored. Here we show that eIF4A1 enhances translational repression during the inhibition of mechanistic target of rapamycin complex 1 (mTORC1), an essential kinase complex controlling cell proliferation. RNA pulldown followed by sequencing revealed that eIF4A1 preferentially binds to mRNAs containing terminal oligopyrimidine (TOP) motifs, whose translation is rapidly repressed upon mTORC1 inhibition. This selective interaction depends on a La-related RNA-binding protein, LARP1. Ribosome profiling revealed that deletion of EIF4A1 attenuated the translational repression of TOP mRNAs upon mTORC1 inactivation. Moreover, eIF4A1 increases the interaction between TOP mRNAs and LARP1 and, thus, ensures stronger translational repression upon mTORC1 inhibition. Our data show the multimodality of eIF4A1 in modulating protein synthesis through an inhibitory binding partner and provide a unique example of the repressive role of a universal translational activator. The authors revealed that the general translation factor eIF4A exerts a repressive effect on a subset of mRNAs by enhancing LARP1 and TOP mRNAs during mTORC1 inhibition under stress.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature Structural & Molecular Biology
BIOCHEMISTRY & MOLECULAR BIOLOGY-BIOPHYSICS
CiteScore
22.00
自引率
1.80%
发文量
160
审稿时长
3-8 weeks
期刊介绍:
Nature Structural & Molecular Biology is a comprehensive platform that combines structural and molecular research. Our journal focuses on exploring the functional and mechanistic aspects of biological processes, emphasizing how molecular components collaborate to achieve a particular function. While structural data can shed light on these insights, our publication does not require them as a prerequisite.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


