塞那帕利作为晚期卵巢癌的一线维持疗法:随机3期试验
IF 50
1区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
摘要
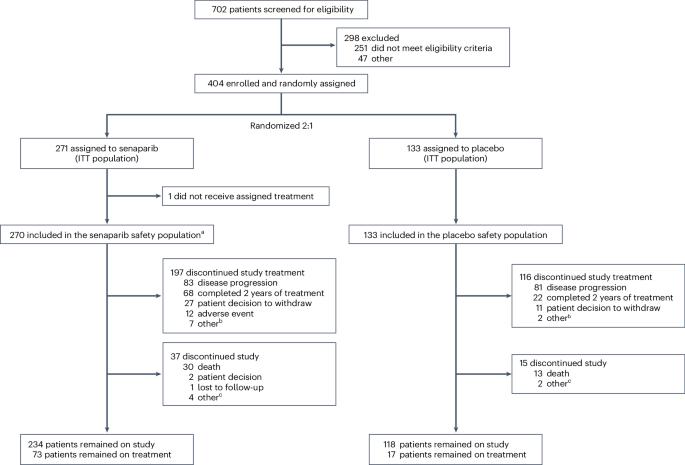
聚腺苷二磷酸核糖聚合酶(PARP)抑制剂作为一线化疗后的维持疗法,提高了晚期卵巢癌女性患者的无进展生存率;然而,并非所有PARP抑制剂都能为未经生物标志物筛选的人群带来益处。Senaparib 是一种 PARP 抑制剂,在 1 期研究中对包括卵巢癌在内的实体瘤患者显示出抗肿瘤活性。多中心、双盲、3 期试验 FLAMES 将 404 名患有晚期卵巢癌(国际妇产科联盟 III-IV 期)并对一线铂类化疗有反应的女性患者随机(2:1)分为塞那帕利 100 毫克(n = 271)或安慰剂(n = 133),每天口服一次,疗程长达 2 年。主要终点是无进展生存期,由盲法独立中央审查评估。在预设的中期分析中,senaparib的中位无进展生存期没有达到,而安慰剂的中位无进展生存期为13.6个月(危险比为0.43,95%置信区间为0.32-0.58;P< 0.0001)。在根据BRCA1和BRCA2突变或同源重组状态定义的亚组中,塞那帕利的获益时间与安慰剂一致。发生≥3级治疗突发不良事件的患者分别为179例(66%)和27例(20%)。与安慰剂相比,塞那帕利能明显改善一线铂类化疗反应后的晚期卵巢癌患者的无进展生存期,与BRCA1和BRCA2突变状态无关,在同源重组亚组之间也能观察到一致的获益,而且耐受性良好。这些结果支持塞那帕利作为一线化疗反应后晚期卵巢癌患者的维持治疗药物。ClinicalTrials.gov identifier:NCT04169997。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Senaparib as first-line maintenance therapy in advanced ovarian cancer: a randomized phase 3 trial
Poly(adenosine diphosphate-ribose) polymerase (PARP) inhibitors as maintenance therapy after first-line chemotherapy have improved progression-free survival in women with advanced ovarian cancer; however, not all PARP inhibitors can provide benefit for a biomarker-unselected population. Senaparib is a PARP inhibitor that demonstrated antitumor activity in patients with solid tumors, including ovarian cancer, in phase 1 studies. The multicenter, double-blind, phase 3 trial FLAMES randomized (2:1) 404 females with advanced ovarian cancer (International Federation of Gynecology and Obstetrics stage III–IV) and response to first-line platinum-based chemotherapy to senaparib 100 mg (n = 271) or placebo (n = 133) orally once daily for up to 2 years. The primary endpoint was progression-free survival assessed by blinded independent central review. At the prespecified interim analysis, the median progression-free survival was not reached with senaparib and was 13.6 months with placebo (hazard ratio 0.43, 95% confidence interval 0.32–0.58; P < 0.0001). The benefit with senaparib over placebo was consistent in the subgroups defined by BRCA1 and BRCA2 mutation or homologous recombination status. Grade ≥3 treatment-emergent adverse events occurred in 179 (66%) and 27 (20%) patients, respectively. Senaparib significantly improved progression-free survival versus placebo in patients with advanced ovarian cancer after response to first-line platinum-based chemotherapy, irrespective of BRCA1 and BRCA2 mutation status and with consistent benefits observed between homologous recombination subgroups, and was well tolerated. These results support senaparib as a maintenance treatment for patients with advanced ovarian cancer after a response to first-line chemotherapy. ClinicalTrials.gov identifier: NCT04169997 . In a prespecified interim analysis of the multicenter, randomized, phase 3 FLAMES trial, maintenance therapy with a PARP inhibitor in patients with ovarian cancer showed prolonged progression-free survival compared with placebo in all subgroups defined by BRCA or homologous recombination status.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature Medicine
医学-生化与分子生物学
CiteScore
100.90
自引率
0.70%
发文量
525
审稿时长
1 months
期刊介绍:
Nature Medicine is a monthly journal publishing original peer-reviewed research in all areas of medicine. The publication focuses on originality, timeliness, interdisciplinary interest, and the impact on improving human health. In addition to research articles, Nature Medicine also publishes commissioned content such as News, Reviews, and Perspectives. This content aims to provide context for the latest advances in translational and clinical research, reaching a wide audience of M.D. and Ph.D. readers. All editorial decisions for the journal are made by a team of full-time professional editors.
Nature Medicine consider all types of clinical research, including:
-Case-reports and small case series
-Clinical trials, whether phase 1, 2, 3 or 4
-Observational studies
-Meta-analyses
-Biomarker studies
-Public and global health studies
Nature Medicine is also committed to facilitating communication between translational and clinical researchers. As such, we consider “hybrid” studies with preclinical and translational findings reported alongside data from clinical studies.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


