尿石素 a 通过激活肿瘤巨噬细胞中由 TFEB 介导的有丝分裂抑制乳腺癌的进展。
IF 11.4
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
摘要
本文章由计算机程序翻译,如有差异,请以英文原文为准。
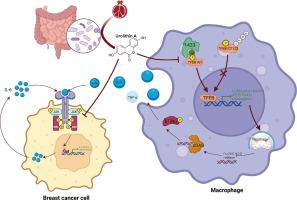

Urolithin A inhibits breast cancer progression via activating TFEB-mediated mitophagy in tumor macrophages
Introduction
Urolithin A (UA) is a naturally occurring compound that is converted from ellagitannin-like precursors in pomegranates and nuts by intestinal flora. Previous studies have found that UA exerts tumor-suppressive effects through antitumor cell proliferation and promotion of memory T-cell expansion, but its role in tumor-associated macrophages remains unknown.
Objectives
Our study aims to reveal how UA affects tumor macrophages and tumor cells to inhibit breast cancer progression.
Methods
Observe the effect of UA treatment on breast cancer progression though in vivo and in vitro experiments. Western blot and PCR assays were performed to discover that UA affects tumor macrophage autophagy and inflammation. Co-ip and Molecular docking were used to explore specific molecular mechanisms.
Results
We observed that UA treatment could simultaneously inhibit harmful inflammatory factors, especially for InterleuKin-6 (IL-6) and tumor necrosis factor α (TNF-α), in both breast cancer cells and tumor-associated macrophages, thereby improving the tumor microenvironment and delaying tumor progression. Mechanistically, UA induced the key regulator of autophagy, transcription factor EB (TFEB), into the nucleus in a partially mTOR-dependent manner and inhibited the ubiquitination degradation of TFEB, which facilitated the clearance of damaged mitochondria via the mitophagy-lysosomal pathway in macrophages under tumor supernatant stress, and reduced the deleterious inflammatory factors induced by the release of nucleic acid from damaged mitochondria. Molecular docking and experimental studies suggest that UA block the recognition of TFEB by 1433 and induce TFEB nuclear localization. Notably, UA treatment demonstrated inhibitory effects on tumor progression in multiple breast cancer models.
Conclusion
Our study elucidated the anti-breast cancer effect of UA from the perspective of tumor-associated macrophages. Specifically, TFEB is a crucial downstream target in macrophages.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Journal of Advanced Research
Multidisciplinary-Multidisciplinary
CiteScore
21.60
自引率
0.90%
发文量
280
审稿时长
12 weeks
期刊介绍:
Journal of Advanced Research (J. Adv. Res.) is an applied/natural sciences, peer-reviewed journal that focuses on interdisciplinary research. The journal aims to contribute to applied research and knowledge worldwide through the publication of original and high-quality research articles in the fields of Medicine, Pharmaceutical Sciences, Dentistry, Physical Therapy, Veterinary Medicine, and Basic and Biological Sciences.
The following abstracting and indexing services cover the Journal of Advanced Research: PubMed/Medline, Essential Science Indicators, Web of Science, Scopus, PubMed Central, PubMed, Science Citation Index Expanded, Directory of Open Access Journals (DOAJ), and INSPEC.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


