人类 Cav3.2 受选择性拮抗剂抑制的结构基础
IF 28.1
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
摘要
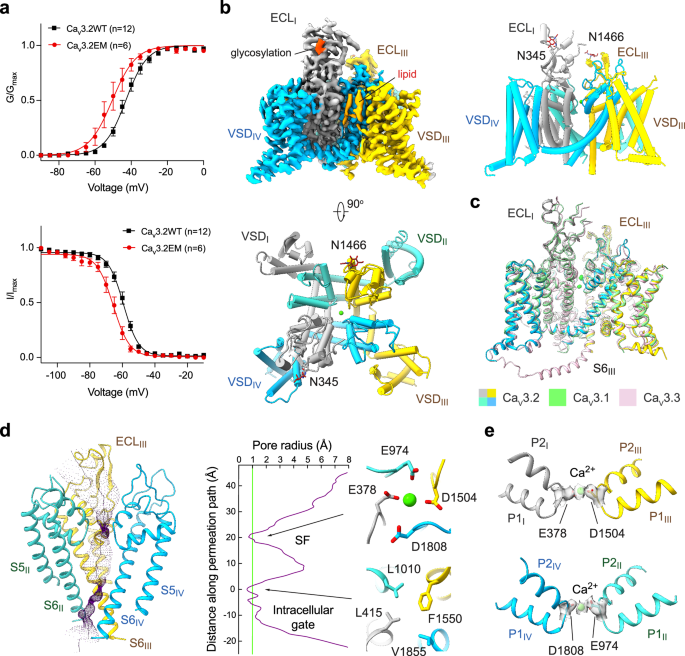
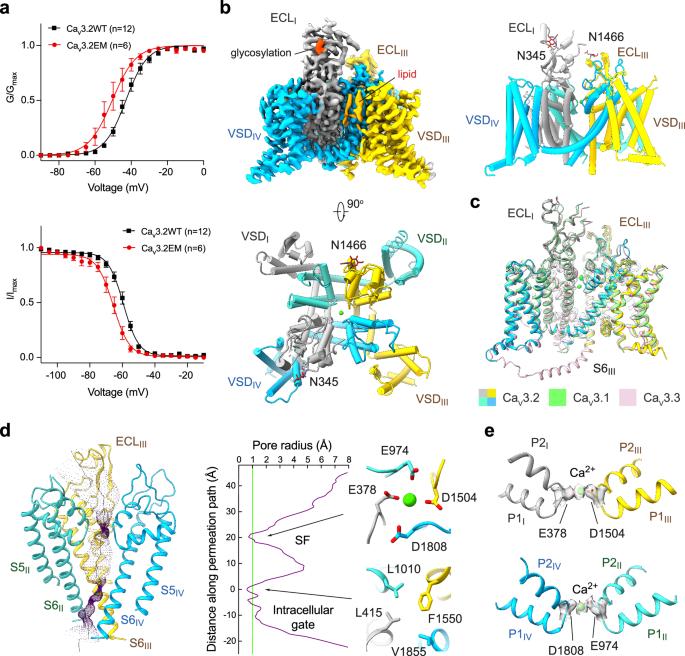
T 型钙通道的 Cav3.2 亚型因其在疼痛和癫痫中的作用而成为开发镇痛药和抗癫痫药的目标。在此,我们展示了 Cav3.2 单独和与四种 T 型钙通道选择性拮抗剂复合物的低温电子显微镜结构,其整体分辨率为 2.8 Å 至 3.2 Å。这四种化合物显示出两种结合姿态。ACT-709478 和 TTA-A2 都将其含环丙基苯基的末端置于中心空腔,直接阻碍离子流动,同时将其极性尾端伸入 IV-I 栅栏。TTA-P2 和 ML218 将其 3,5-二氯苯甲酰胺基团伸入 II-III 级栅栏,并将其疏水尾部置于空腔中,以阻碍离子渗透。栅栏穿透模式立即解释了这些拮抗剂依赖于状态的活性。结构引导突变分析确定了决定这些药物 T 型偏好的几个关键残基。这些结构还表明了一种内源性脂质在稳定药物在中心腔的结合方面所起的作用。本文章由计算机程序翻译,如有差异,请以英文原文为准。
Structural basis for human Cav3.2 inhibition by selective antagonists
The Cav3.2 subtype of T-type calcium channels has been targeted for developing analgesics and anti-epileptics for its role in pain and epilepsy. Here we present the cryo-EM structures of Cav3.2 alone and in complex with four T-type calcium channel selective antagonists with overall resolutions ranging from 2.8 Å to 3.2 Å. The four compounds display two binding poses. ACT-709478 and TTA-A2 both place their cyclopropylphenyl-containing ends in the central cavity to directly obstruct ion flow, meanwhile extending their polar tails into the IV-I fenestration. TTA-P2 and ML218 project their 3,5-dichlorobenzamide groups into the II-III fenestration and place their hydrophobic tails in the cavity to impede ion permeation. The fenestration-penetrating mode immediately affords an explanation for the state-dependent activities of these antagonists. Structure-guided mutational analysis identifies several key residues that determine the T-type preference of these drugs. The structures also suggest the role of an endogenous lipid in stabilizing drug binding in the central cavity.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Cell Research
生物-细胞生物学
CiteScore
53.90
自引率
0.70%
发文量
2420
审稿时长
2.3 months
期刊介绍:
Cell Research (CR) is an international journal published by Springer Nature in partnership with the Center for Excellence in Molecular Cell Science, Chinese Academy of Sciences (CAS). It focuses on publishing original research articles and reviews in various areas of life sciences, particularly those related to molecular and cell biology. The journal covers a broad range of topics including cell growth, differentiation, and apoptosis; signal transduction; stem cell biology and development; chromatin, epigenetics, and transcription; RNA biology; structural and molecular biology; cancer biology and metabolism; immunity and molecular pathogenesis; molecular and cellular neuroscience; plant molecular and cell biology; and omics, system biology, and synthetic biology. CR is recognized as China's best international journal in life sciences and is part of Springer Nature's prestigious family of Molecular Cell Biology journals.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


