银和金介导的三氟甲基化反应的基本步骤
IF 2.5
3区 化学
Q2 CHEMISTRY, INORGANIC & NUCLEAR
引用次数: 0
摘要
由于三氟甲基在药物化学中的重要性,三氟甲基化反应备受青睐。大多数已报道的三氟甲基化反应方案都依赖于铜络合物,而铜络合物在这些转化中的机理作用已被广泛分析。有初步证据表明,银和金配合物也有可能促成三氟甲基化反应,但对这些过程仍缺乏更详细的机理了解。在这里,我们使用电喷雾电离质谱法研究了溶液中 [Ag(R)(CF3)3]- 和 [Au(R)(CF3)3]- (R = Me、Et、nBu、sBu、烯丙基、芳基)的形成,并通过气相碎片实验和量子化学计算描述了它们的单分子反应性。精氨酸络合物既会发生 RCF3 的协同还原消除,也会发生 R- 和 CF3- 的双倍自由基损失。与类似的铜酸盐络合物一样,两种相互竞争的破碎途径之间的分支比例受 R- 自由基稳定性的控制。相比之下,金酸盐的相应碎片反应的计算障碍要高得多,因此这些配合物通过其他途径解离,不会产生 RCF3 产物。本文章由计算机程序翻译,如有差异,请以英文原文为准。
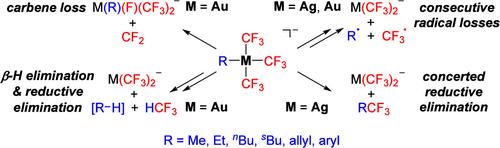
Elementary Steps of Silver- and Gold-Mediated Trifluoromethylation Reactions
Due to the importance of the trifluoromethyl group in medicinal chemistry, trifluoromethylation reactions are in high demand. Most of the reported trifluoromethylation protocols rely on copper complexes, whose mechanistic role in these transformations has been analyzed extensively. First evidence also points to the potential of silver and gold complexes for enabling trifluoromethylation reactions, but a more detailed mechanistic understanding of these processes is still lacking. Here, we use electrospray-ionization mass spectrometry to study the formation of [Ag(R)(CF3)3]− and [Au(R)(CF3)3]− (R = Me, Et, nBu, sBu, allyl, aryl) in solution and characterize their unimolecular reactivity by gas-phase fragmentation experiments and quantum chemical calculations. The argentate complexes undergo both concerted reductive eliminations of RCF3 and 2-fold radical losses of R• and CF3•. Like in the case of the analogous cuprate complexes, the branching ratio between the two competing fragmentation channels is controlled by the stability of the R• radicals. In contrast, the calculated barriers of the corresponding fragmentation reactions of the aurates are much higher, for which reason these complexes dissociate by alternative pathways and do not afford the RCF3 products.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Organometallics
化学-无机化学与核化学
CiteScore
5.60
自引率
7.10%
发文量
382
审稿时长
1.7 months
期刊介绍:
Organometallics is the flagship journal of organometallic chemistry and records progress in one of the most active fields of science, bridging organic and inorganic chemistry. The journal publishes Articles, Communications, Reviews, and Tutorials (instructional overviews) that depict research on the synthesis, structure, bonding, chemical reactivity, and reaction mechanisms for a variety of applications, including catalyst design and catalytic processes; main-group, transition-metal, and lanthanide and actinide metal chemistry; synthetic aspects of polymer science and materials science; and bioorganometallic chemistry.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


