设计和优化蛋白质功能的机遇与挑战
IF 81.3
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
摘要
过去十年,蛋白质设计领域取得了显著进展。过去,纯粹基于结构的设计方法可靠性低,限制了它们的应用,但最近结合了基于结构和基于序列计算的策略以及机器学习工具,极大地改进了蛋白质工程和设计。在这篇综述中,我们将讨论这些方法是如何帮助设计出日益复杂的结构和治疗相关活性的。此外,蛋白质优化方法还提高了复杂真核蛋白质的稳定性和活性。由于可靠性的提高,计算设计方法已被用于改进绿色化学疗法和酶,并产生了疫苗抗原、抗病毒药物和给药纳米载体。此外,设计方法的巨大成功也反映出人们对蛋白质序列、结构和功能之间关系的基本规律有了更深入的了解。然而,从头设计仍主要局限于α螺旋束,限制了其生成复杂酶和多种蛋白质及小分子结合剂的潜力。如果我们要实现产生新的自然活性的目标,设计复杂的蛋白质结构是具有挑战性但又是必要的下一步。本文章由计算机程序翻译,如有差异,请以英文原文为准。
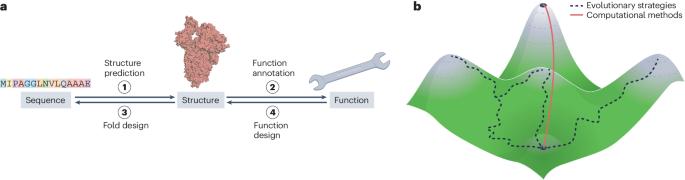
Opportunities and challenges in design and optimization of protein function
The field of protein design has made remarkable progress over the past decade. Historically, the low reliability of purely structure-based design methods limited their application, but recent strategies that combine structure-based and sequence-based calculations, as well as machine learning tools, have dramatically improved protein engineering and design. In this Review, we discuss how these methods have enabled the design of increasingly complex structures and therapeutically relevant activities. Additionally, protein optimization methods have improved the stability and activity of complex eukaryotic proteins. Thanks to their increased reliability, computational design methods have been applied to improve therapeutics and enzymes for green chemistry and have generated vaccine antigens, antivirals and drug-delivery nano-vehicles. Moreover, the high success of design methods reflects an increased understanding of basic rules that govern the relationships among protein sequence, structure and function. However, de novo design is still limited mostly to α-helix bundles, restricting its potential to generate sophisticated enzymes and diverse protein and small-molecule binders. Designing complex protein structures is a challenging but necessary next step if we are to realize our objective of generating new-to-nature activities. Recent combinations of structure-based and sequence-based calculations and machine learning tools have dramatically improved protein engineering and design. Although designing complex protein structures remains challenging, these methods have enabled the design of therapeutically relevant activities, including vaccine antigens, antivirals and drug-delivery nano-vehicles.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
173.60
自引率
0.50%
发文量
118
审稿时长
6-12 weeks
期刊介绍:
Nature Reviews Molecular Cell Biology is a prestigious journal that aims to be the primary source of reviews and commentaries for the scientific communities it serves. The journal strives to publish articles that are authoritative, accessible, and enriched with easily understandable figures, tables, and other display items. The goal is to provide an unparalleled service to authors, referees, and readers, and the journal works diligently to maximize the usefulness and impact of each article. Nature Reviews Molecular Cell Biology publishes a variety of article types, including Reviews, Perspectives, Comments, and Research Highlights, all of which are relevant to molecular and cell biologists. The journal's broad scope ensures that the articles it publishes reach the widest possible audience.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


