小分子原药的前景
IF 122.7
1区 医学
Q1 BIOTECHNOLOGY & APPLIED MICROBIOLOGY
引用次数: 0
摘要
原研药是具有优于母体活性药物成分(API)特性的衍生物,服用后经过生物转化在原位生成 API。虽然原研药具有这一普遍特征,但其化学结构、治疗适应症和特性却千差万别。在此,我们首次利用化学信息学和数据科学方法对目前已批准的原研药进行了整体分析,以揭示原研药的开发趋势。我们强调了原药设计的基本原理、适应症、原料药释放机制、添加到原料药中形成原药的原药化学以及原药对市场的影响。在此分析的基础上,我们讨论了当前原药方法的优势和局限性,并提出了未来的发展领域。本文章由计算机程序翻译,如有差异,请以英文原文为准。
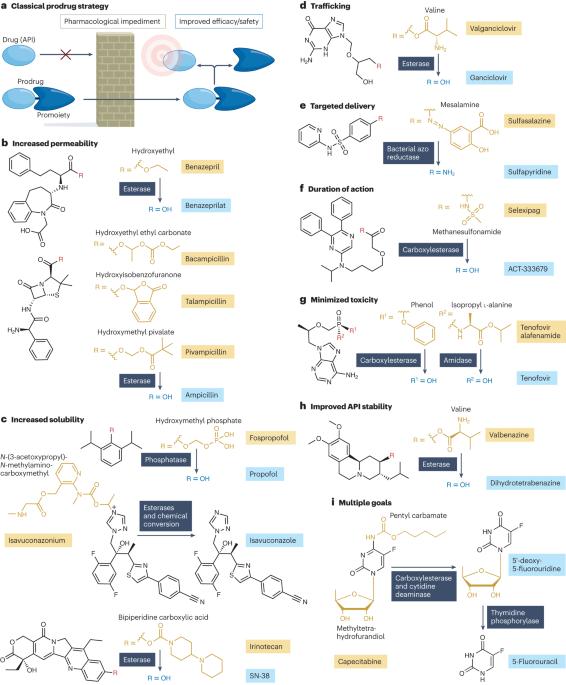
The landscape of small-molecule prodrugs
Prodrugs are derivatives with superior properties compared with the parent active pharmaceutical ingredient (API), which undergo biotransformation after administration to generate the API in situ. Although sharing this general characteristic, prodrugs encompass a wide range of different chemical structures, therapeutic indications and properties. Here we provide the first holistic analysis of the current landscape of approved prodrugs using cheminformatics and data science approaches to reveal trends in prodrug development. We highlight rationales that underlie prodrug design, their indications, mechanisms of API release, the chemistry of promoieties added to APIs to form prodrugs and the market impact of prodrugs. On the basis of this analysis, we discuss strengths and limitations of current prodrug approaches and suggest areas for future development. The development of prodrugs — derivatives of active pharmaceutical ingredients (APIs) with little or no biological activity themselves that are converted into the API after administration — can address issues with properties of the API such as poor bioavailability. This article provides a holistic analysis of approved prodrugs and discusses trends in prodrug design, their indications, mechanisms of API release and the chemistry of promoieties added to APIs to form prodrugs.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Nature Reviews. Drug Discovery
医学-生物工程与应用微生物
CiteScore
137.40
自引率
0.30%
发文量
227
期刊介绍:
Nature Reviews Drug Discovery is a monthly journal aimed at everyone working in the drug discovery and development arena.
Each issue includes:
Highest-quality reviews and perspectives covering a broad scope.
News stories investigating the hottest topics in drug discovery.
Timely summaries of key primary research papers.
Concise updates on the latest advances in areas such as new drug approvals, patent law, and emerging industry trends and strategies.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


