表皮生长因子受体靶向疗法:临床活性、抗药性机制和新方向。
IF 81.1
1区 医学
Q1 ONCOLOGY
引用次数: 0
摘要
成纤维细胞生长因子(FGF)信号通过 FGF 受体(FGFR1-4)协调胎儿发育,促进组织和全身平衡,但也可促进肿瘤发生。目前已开发出多种药物,包括泛FGFR抑制剂(厄达非替尼和福替巴替尼)、FGFR1/2/3抑制剂(infigratinib和pemigatinib)以及一系列更具特异性的药物,其中一些已进入临床应用。Erdafitinib 已获准用于治疗携带 FGFR2/3 基因改变的尿路上皮癌患者,futibatinib 和 pemigatinib 已获准用于治疗携带 FGFR2 融合和/或重排的胆管癌患者。由于 FGFR1 的脱靶抑制、FGFR 基因的耐药突变、旁路信号通路的激活、同时发生的 TP53 改变以及可能与上皮-间质转化相关的同工酶转换,这些药物的临床获益部分受到高磷血症的限制。下一代小分子抑制剂(如 lirafugratinib 和 LOXO-435)和 FGFR2 特异性抗体 bemarituzumab 预计将降低高磷血症的风险,并能克服某些耐药突变。在本综述中,我们将介绍 FGFR 抑制剂的发展和目前的临床作用,并展望未来的研究方向,包括扩大 FGFR 抑制剂的治疗适应症、将这些药物与免疫检查点抑制剂联合使用以及应用人工智能等新技术。本文章由计算机程序翻译,如有差异,请以英文原文为准。
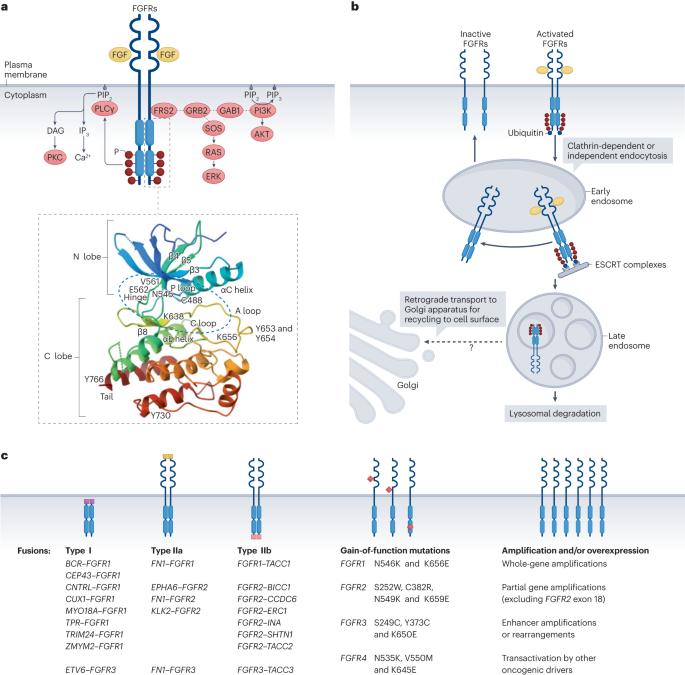
FGFR-targeted therapeutics: clinical activity, mechanisms of resistance and new directions
Fibroblast growth factor (FGF) signalling via FGF receptors (FGFR1–4) orchestrates fetal development and contributes to tissue and whole-body homeostasis, but can also promote tumorigenesis. Various agents, including pan-FGFR inhibitors (erdafitinib and futibatinib), FGFR1/2/3 inhibitors (infigratinib and pemigatinib), as well as a range of more-specific agents, have been developed and several have entered clinical use. Erdafitinib is approved for patients with urothelial carcinoma harbouring FGFR2/3 alterations, and futibatinib and pemigatinib are approved for patients with cholangiocarcinoma harbouring FGFR2 fusions and/or rearrangements. Clinical benefit from these agents is in part limited by hyperphosphataemia owing to off-target inhibition of FGFR1 as well as the emergence of resistance mutations in FGFR genes, activation of bypass signalling pathways, concurrent TP53 alterations and possibly epithelial–mesenchymal transition-related isoform switching. The next generation of small-molecule inhibitors, such as lirafugratinib and LOXO-435, and the FGFR2-specific antibody bemarituzumab are expected to have a reduced risk of hyperphosphataemia and the ability to overcome certain resistance mutations. In this Review, we describe the development and current clinical role of FGFR inhibitors and provide perspective on future research directions including expansion of the therapeutic indications for use of FGFR inhibitors, combination of these agents with immune-checkpoint inhibitors and the application of novel technologies, such as artificial intelligence. FGFR inhibitors are now approved for use in patients with advanced-stage urothelial carcinoma, cholangiocarcinoma and myeloid or lymphoid neoplasms that harbour certain FGFR alterations. Nonetheless, challenges such as tolerability and acquired resistance limit the clinical potential of these agents. In this Review, the authors summarize the available clinical data on FGFR inhibitors, describe promising novel agents and highlight future research directions that might optimize the efficacy of FGFR-targeted therapies.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊
CiteScore
99.40
自引率
0.40%
发文量
114
审稿时长
6-12 weeks
期刊介绍:
Nature Reviews publishes clinical content authored by internationally renowned clinical academics and researchers, catering to readers in the medical sciences at postgraduate levels and beyond. Although targeted at practicing doctors, researchers, and academics within specific specialties, the aim is to ensure accessibility for readers across various medical disciplines. The journal features in-depth Reviews offering authoritative and current information, contextualizing topics within the history and development of a field. Perspectives, News & Views articles, and the Research Highlights section provide topical discussions, opinions, and filtered primary research from diverse medical journals.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


