苄基 C(sp3)-H 氮化:铜催化与铁催化
IF 17.7
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
摘要
通过氢原子抽取(HAT)生成苄基自由基是近年来的研究重点,各种 C(sp3)-H 键官能化方案都是基于这一基本步骤开发的。我们在此报告铜催化和铁催化的 C(sp3)-H 苄基叠氮反应,分别使用 mCPBA 和 NFSI 作为氧化剂,TMSN3 作为叠氮源。该反应被认为是通过原位生成的以杂原子为中心的自由基在分子间抽取苄基氢而引发的。Fe(OTf)3 催化的叠氮化反应具有良好的化学选择性,因为它优先发生在二级和三级苄基 C(sp3)-H 键上,而不是一级苄基和三级脂肪族碳上。此外,还记录了开发催化对映体选择性工艺的努力。本文章由计算机程序翻译,如有差异,请以英文原文为准。
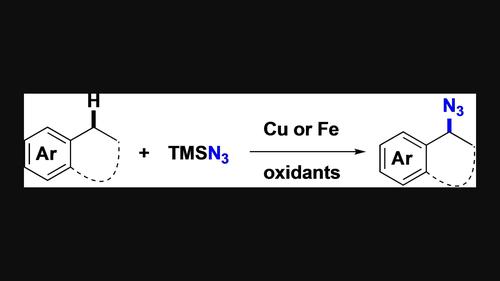
Benzylic C(sp3)‐H Azidation: Copper vs Iron Catalysis
The generation of benzylic radicals through hydrogen atom abstraction (HAT) has been a recent research focus and various C(sp3)‐H bond functionalization protocols have been developed relying on this elementary step. We report herein copper‐ and iron‐catalyzed C(sp3)‐H benzylic azidation reactions using mCPBA and NFSI as oxidant, respectively, and TMSN3 as azide source. The reaction is thought to be initiated via intermolecular abstraction of benzylic hydrogen by the in situ generated heteroatom‐centered radicals. The Fe(OTf)3‐catalyzed azidation protocol displays good chemoselectivity as it takes place preferentially at the secondary and tertiary benzylic C(sp3)‐H bonds over the primary benzylic and tertiary aliphatic carbons. Efforts on the development of catalytic enantioselective processes are also documented.
求助全文
通过发布文献求助,成功后即可免费获取论文全文。
去求助
来源期刊

Accounts of Chemical Research
化学-化学综合
CiteScore
31.40
自引率
1.10%
发文量
312
审稿时长
2 months
期刊介绍:
Accounts of Chemical Research presents short, concise and critical articles offering easy-to-read overviews of basic research and applications in all areas of chemistry and biochemistry. These short reviews focus on research from the author’s own laboratory and are designed to teach the reader about a research project. In addition, Accounts of Chemical Research publishes commentaries that give an informed opinion on a current research problem. Special Issues online are devoted to a single topic of unusual activity and significance.
Accounts of Chemical Research replaces the traditional article abstract with an article "Conspectus." These entries synopsize the research affording the reader a closer look at the content and significance of an article. Through this provision of a more detailed description of the article contents, the Conspectus enhances the article's discoverability by search engines and the exposure for the research.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:



