Added value of molecular karyotype in childhood acute lymphoblastic leukemia
Abstract
Background
Thanks to an improved therapeutic regimen in childhood B-cell precursor acute lymphoblastic leukemia (BCP-ALL), 5 year-overall survival now exceeds 90%. Unfortunately, the 25% of children who relapse have an initial poor prognosis, potentially driven by pre-existing or emerging molecular anomalies. The latter are initially and essentially identified by cytogenetics. However, some subtle alterations are not visible through karyotyping.
Methods
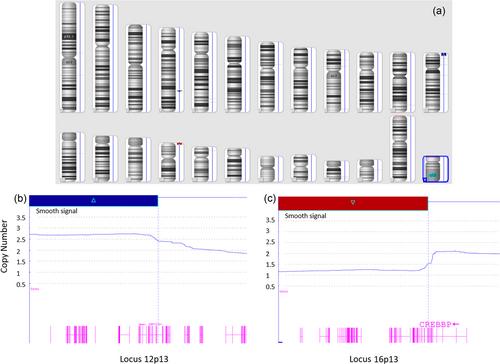
Single nucleotide polymorphisms (SNP) array is an alternative way of chromosomal analysis allowing for a more in-depth evaluation of chromosomal modifications such as the assessment of copy number alterations (CNA) and loss of heterozygosity (LOH). This method was applied here in retrospective diagnosis/relapse paired samples from seven children with BCP-ALL and in a prospective cohort of 38 newly diagnosed childhood cases.
Results
In the matched study, compared to the initial karyotype, SNP array analysis reclassified two patients as poor prognosis cases. Modulation during relapse was seen for 4 CNA and 0.9 LOH. In the prospective study, SNP reclassified the 10 patients with intermediate karyotype as 7 good prognosis and 3 poor prognosis. Ultimately, in all the children tested, SNP array allowed to identify additional anomalies compared to conventional karyotype, refine its prognostic value and identify some druggable anomalies that could be used for precision medicine. Overall, the anomalies detected could be segregated in four groups respectively involved in B-cell development, cell proliferation, transcription and molecular pathways.
Conclusion
SNP therefore appears to be a method of choice in the integrated diagnosis of BCP ALL, especially for patients initially classified as intermediate prognosis. This complementary method of both cytogenetics and high throughput sequencing allows to obtain further classified information and can be useful in case of failure of these techniques.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


