Sustained activation of persulfate by slow release of Fe(II) from silica-coated nanosized zero-valent iron for in situ chemical oxidation
Abstract
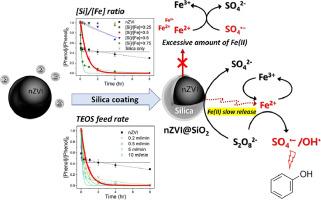
Sustained activation of persulfate through the slow release of Fe(II) from silica-coated nanosized zero-valent iron (nZVI) particles (nZVI@SiO2) was investigated. Slow release of Fe(II) prevented radical scavenging by excess Fe(II) and increased the radical yield, which improved the stoichiometric efficiency of phenol degradation. Sulfate and hydroxyl radicals were found to be the main oxidative species produced during phenol degradation and were found to make comparable contributions to oxidation. The nZVI@SiO2 particle silica shell thickness controlled the release of Fe(II) and therefore the sustained activation of persulfate and was strongly affected by the synthesis conditions, including the [Si]/[Fe] ratio and silica supply rate. Optimal sustained phenol degradation was achieved when nZVI@SiO2 particles were synthesized using a [Si]/[Fe] ratio of 0.5 and a tetraethyl orthosilicate supply rate of 0.5 mL/min, and this was attributed to the nZVI@SiO2 particles giving an optimal Fe(II) release rate and therefore a high persulfate activation rate and a high phenol removal efficiency. Sustained persulfate activation induced by Fe(II) being slowly released was described well by single-stage first-order kinetics rather than two-stage first-order kinetics typical of unmodified nZVI/persulfate systems. Persulfate was found still to be activated by iron (oxyhydr)oxides minerals after the nZVI@SiO2 particles had been exhausted but the persulfate sustained activation induced by the slow release of Fe(II) played a crucial role in determining the overall degradation efficiency. The results highlight the importance of the slow release of Fe(II) from nZVI-based materials for in situ chemical oxidation through sustained persulfate activation.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


