Optimizing the p charge of S in p-block metal sulfides for sulfur reduction electrocatalysis
IF 42.8
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 13
Abstract
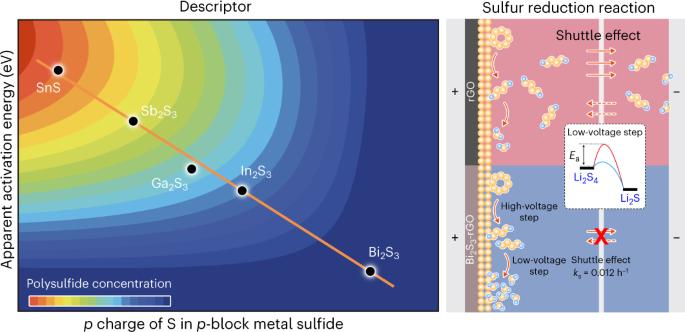
Understanding sulfur conversion chemistry is key to the development of sulfur-based high-energy-density batteries. However, unclear relationships between the electronic structure of the catalyst and its activity are the major problem. Here, we provide a direct correlation between the p electron gain of S in p-block metal sulfides and the apparent activation energies (Ea) for the sulfur reduction reaction (SRR), in particular, Li2Sn to Li2S conversion, which is the rate-determining step of the SRR. The maximum p charge occurs in bismuth sulfide and results in the lowest Ea and a high SRR rate in the cathode. Li–S batteries with the Bi2S3 catalyst work steadily at a high rate of 5.0C with a high-capacity retention of ~85% after 500 cycles. A high areal capacity of ~21.9 mAh cm−2 was obtained under a high sulfur loading of 17.6 mg cm−2 but a low electrolyte/sulfur ratio of 7.5 μl mg−1. Lithium–sulfur batteries are promising energy storage devices where catalysis can play an important role, but developing design principles for optimal performance remains a challenge. Now, a series of p-block metal sulfide cathodes are evaluated, revealing a direct correlation between the p electron gain of sulfur in the sulfide material and the apparent activation energy for the sulfur reduction reaction.
优化p块金属硫化物中硫还原电催化的p电荷
了解硫转化化学是开发硫基高能量密度电池的关键。然而,催化剂的电子结构与其活性之间的关系不明确是一个主要问题。在这里,我们提供了 p 嵌段金属硫化物中 S 的 p 电子增益与硫还原反应(SRR)表观活化能(Ea)之间的直接相关性,特别是 Li2Sn 到 Li2S 的转化,这是 SRR 的决定性步骤。硫化铋中的 p 电荷最大,因此 Ea 最低,阴极的 SRR 速率较高。使用 Bi2S3 催化剂的锂-S 电池能在 5.0C 的高速率下稳定工作,循环 500 次后仍能保持约 85% 的高容量。在 17.6 mg cm-2 的高硫负荷和 7.5 μl mg-1 的低电解质/硫比率条件下,获得了约 21.9 mAh cm-2 的高电容。锂硫电池是一种前景广阔的储能设备,催化在其中可以发挥重要作用,但要开发出性能最优的设计原理仍是一项挑战。现在,我们对一系列对嵌段金属硫化物阴极进行了评估,发现硫化物材料中硫的对电子增益与硫还原反应的表观活化能之间存在直接关联。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Catalysis
Chemical Engineering-Bioengineering
CiteScore
52.10
自引率
1.10%
发文量
140
期刊介绍:
Nature Catalysis serves as a platform for researchers across chemistry and related fields, focusing on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, encompassing both fundamental and applied studies. With a particular emphasis on advancing sustainable industries and processes, the journal provides comprehensive coverage of catalysis research, appealing to scientists, engineers, and researchers in academia and industry.
Maintaining the high standards of the Nature brand, Nature Catalysis boasts a dedicated team of professional editors, rigorous peer-review processes, and swift publication times, ensuring editorial independence and quality. The journal publishes work spanning heterogeneous catalysis, homogeneous catalysis, and biocatalysis, covering areas such as catalytic synthesis, mechanisms, characterization, computational studies, nanoparticle catalysis, electrocatalysis, photocatalysis, environmental catalysis, asymmetric catalysis, and various forms of organocatalysis.
文献相关原料
| 公司名称 | 产品信息 | 采购帮参考价格 |
|---|
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


