Pleconaril and ribavirin in new-onset type 1 diabetes: a phase 2 randomized trial
IF 58.7
1区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 0
Abstract
Previous studies showed a low-grade enterovirus infection in the pancreatic islets of patients with newly diagnosed type 1 diabetes (T1D). In the Diabetes Virus Detection (DiViD) Intervention, a phase 2, placebo-controlled, randomized, parallel group, double-blind trial, 96 children and adolescents (aged 6–15 years) with new-onset T1D received antiviral treatment with pleconaril and ribavirin (n = 47) or placebo (n = 49) for 6 months, with the aim of preserving β cell function. The primary endpoint was the mean stimulated C-peptide area under the curve (AUC) 12 months after the initiation of treatment (less than 3 weeks after diagnosis) using a mixed linear model. The model used longitudinal log-transformed serum C-peptide AUCs at baseline, at 3 months, 6 months and 1 year. The primary endpoint was met with the serum C-peptide AUC being higher in the pleconaril and ribavirin treatment group compared to the placebo group at 12 months (average marginal effect = 0.057 in the linear mixed model; 95% confidence interval = 0.004–0.11, P = 0.037). The treatment was well tolerated. The results show that antiviral treatment may preserve residual insulin production in children and adolescent with new-onset T1D. This provides a rationale for further evaluating antiviral strategies in the prevention and treatment of T1D. European Union Drug Regulating Authorities Clinical Trials identifier: 2015-003350-41 . Results from the DiViD Intervention, a phase 2 randomized, placebo-controlled trial, showed that antiviral treatment with pleconaril and ribavirin for 6 months resulted in higher endogenous insulin production in children and adolescents with new-onset type 1 diabetes.
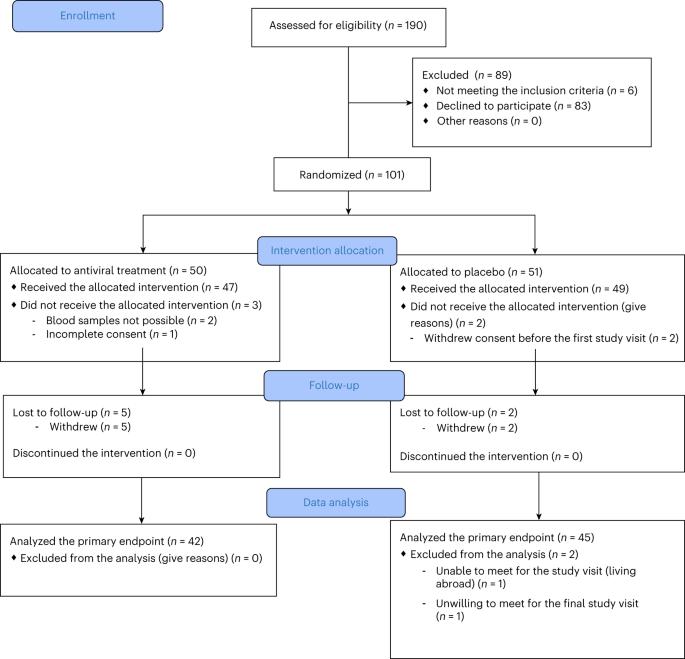
Pleconaril和利巴韦林治疗新发1型糖尿病:一项2期随机试验。
先前的研究表明,新诊断的1型糖尿病(T1D)患者的胰岛中存在低度肠道病毒感染。糖尿病病毒检测(DiViD)干预是一项2期、安慰剂对照、随机、平行组、双盲试验,针对96名6-15岁的儿童和青少年 年)新发T1D患者接受了普勒那立和利巴韦林的抗病毒治疗(n = 47)或安慰剂(n = 49)用于6 月,目的是保留β细胞功能。主要终点是平均刺激的C肽曲线下面积(AUC)12 开始治疗后数月(少于3 诊断后数周)。该模型使用了基线时、3 月,6 月和1 年主要终点是在12岁时,普勒那立和利巴韦林治疗组的血清C肽AUC高于安慰剂组 月(平均边际效应 = 线性混合模型为0.057;95%置信区间 = 0.004-0.11,P = 0.037)。治疗耐受性良好。结果表明,抗病毒治疗可以保留儿童和青少年新发T1D患者的残余胰岛素产生。这为进一步评估预防和治疗T1D的抗病毒策略提供了依据。欧盟药品监管机构临床试验标识符:2015-003350-41。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Medicine
医学-生化与分子生物学
CiteScore
100.90
自引率
0.70%
发文量
525
审稿时长
1 months
期刊介绍:
Nature Medicine is a monthly journal publishing original peer-reviewed research in all areas of medicine. The publication focuses on originality, timeliness, interdisciplinary interest, and the impact on improving human health. In addition to research articles, Nature Medicine also publishes commissioned content such as News, Reviews, and Perspectives. This content aims to provide context for the latest advances in translational and clinical research, reaching a wide audience of M.D. and Ph.D. readers. All editorial decisions for the journal are made by a team of full-time professional editors.
Nature Medicine consider all types of clinical research, including:
-Case-reports and small case series
-Clinical trials, whether phase 1, 2, 3 or 4
-Observational studies
-Meta-analyses
-Biomarker studies
-Public and global health studies
Nature Medicine is also committed to facilitating communication between translational and clinical researchers. As such, we consider “hybrid” studies with preclinical and translational findings reported alongside data from clinical studies.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


