Elranatamab in relapsed or refractory multiple myeloma: the MagnetisMM-1 phase 1 trial
IF 58.7
1区 医学
Q1 BIOCHEMISTRY & MOLECULAR BIOLOGY
引用次数: 5
Abstract
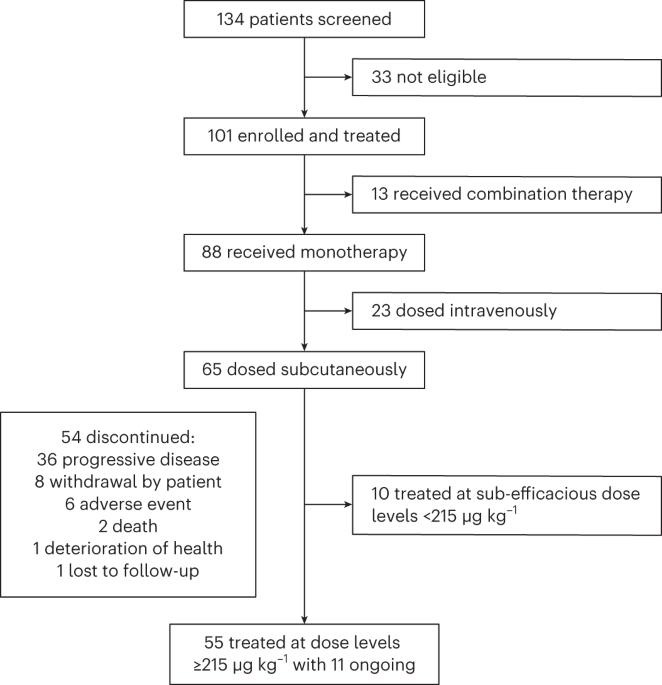
Multiple myeloma (MM) is a plasma cell malignancy expressing B cell maturation antigen (BCMA). Elranatamab, a bispecific antibody, engages BCMA on MM and CD3 on T cells. The MagnetisMM-1 trial evaluated its safety, pharmacokinetics and efficacy. Primary endpoints, including the incidence of dose-limiting toxicities as well as objective response rate (ORR) and duration of response (DOR), were met. Secondary efficacy endpoints included progression-free survival (PFS) and overall survival (OS). Eighty-eight patients with relapsed or refractory MM received elranatamab monotherapy, and 55 patients received elranatamab at efficacious doses. Patients had received a median of five prior regimens; 90.9% were triple-class refractory, 29.1% had high cytogenetic risk and 23.6% received prior BCMA-directed therapy. No dose-limiting toxicities were observed during dose escalation. Adverse events included cytopenias and cytokine release syndrome. Exposure was dose proportional. With a median follow-up of 12.0 months, the ORR was 63.6% and 38.2% of patients achieving complete response or better. For responders, the median DOR was 17.1 months. All 13 patients evaluable for minimal residual disease achieved negativity. Even after prior BCMA-directed therapy, 53.8% achieved response. For all 55 patients, median PFS was 11.8 months, and median OS was 21.2 months. Elranatamab achieved durable responses, manageable safety and promising survival for patients with MM. ClinicalTrials.gov Identifier: NCT03269136 . In the first-in-human trial of elranatamab, patients with refractory or relapsed multiple myeloma who received the bispecific antibody against BCMA and CD3 experienced no dose-limiting toxicities during dose escalation, and the agent showed promising clinical efficacy.
Elranatamab治疗复发或难治性多发性骨髓瘤:MagnetisM-1 1期试验。
多发性骨髓瘤(MM)是一种表达B细胞成熟抗原(BCMA)的浆细胞恶性肿瘤。Elranatamab是一种双特异性抗体,与MM上的BCMA和T细胞上的CD3结合。MagneticsMM-1试验评估了其安全性、药代动力学和疗效。达到了主要终点,包括剂量限制性毒性的发生率以及客观反应率(ORR)和反应持续时间(DOR)。次要疗效终点包括无进展生存期(PFS)和总生存期(OS)。88名复发或难治性MM患者接受了elranatamab单药治疗,55名患者接受了有效剂量的elranata单抗治疗。患者之前接受过五种治疗方案的中位数;90.9%为三级难治性,29.1%具有高细胞遗传学风险,23.6%接受过BCMA指导的治疗。在剂量递增过程中未观察到剂量限制毒性。不良事件包括细胞减少和细胞因子释放综合征。暴露与剂量成正比。中位随访12.0 月,ORR分别为63.6%和38.2%的患者达到完全缓解或更好。对于应答者,DOR中位数为17.1 月。所有13名可评估为最小残留疾病的患者均为阴性。即使在先前BCMA指导的治疗后,53.8%的患者仍有反应。对于所有55名患者,中位PFS为11.8 月,中位OS为21.2 月。Elranatamab为MM患者实现了持久的反应、可控的安全性和有希望的生存。ClinicalTrials.gov标识符:NCT03269136。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature Medicine
医学-生化与分子生物学
CiteScore
100.90
自引率
0.70%
发文量
525
审稿时长
1 months
期刊介绍:
Nature Medicine is a monthly journal publishing original peer-reviewed research in all areas of medicine. The publication focuses on originality, timeliness, interdisciplinary interest, and the impact on improving human health. In addition to research articles, Nature Medicine also publishes commissioned content such as News, Reviews, and Perspectives. This content aims to provide context for the latest advances in translational and clinical research, reaching a wide audience of M.D. and Ph.D. readers. All editorial decisions for the journal are made by a team of full-time professional editors.
Nature Medicine consider all types of clinical research, including:
-Case-reports and small case series
-Clinical trials, whether phase 1, 2, 3 or 4
-Observational studies
-Meta-analyses
-Biomarker studies
-Public and global health studies
Nature Medicine is also committed to facilitating communication between translational and clinical researchers. As such, we consider “hybrid” studies with preclinical and translational findings reported alongside data from clinical studies.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


