Triterpenoids manipulate a broad range of virus-host fusion via wrapping the HR2 domain prevalent in viral envelopes
IF 11.7
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 52
Abstract
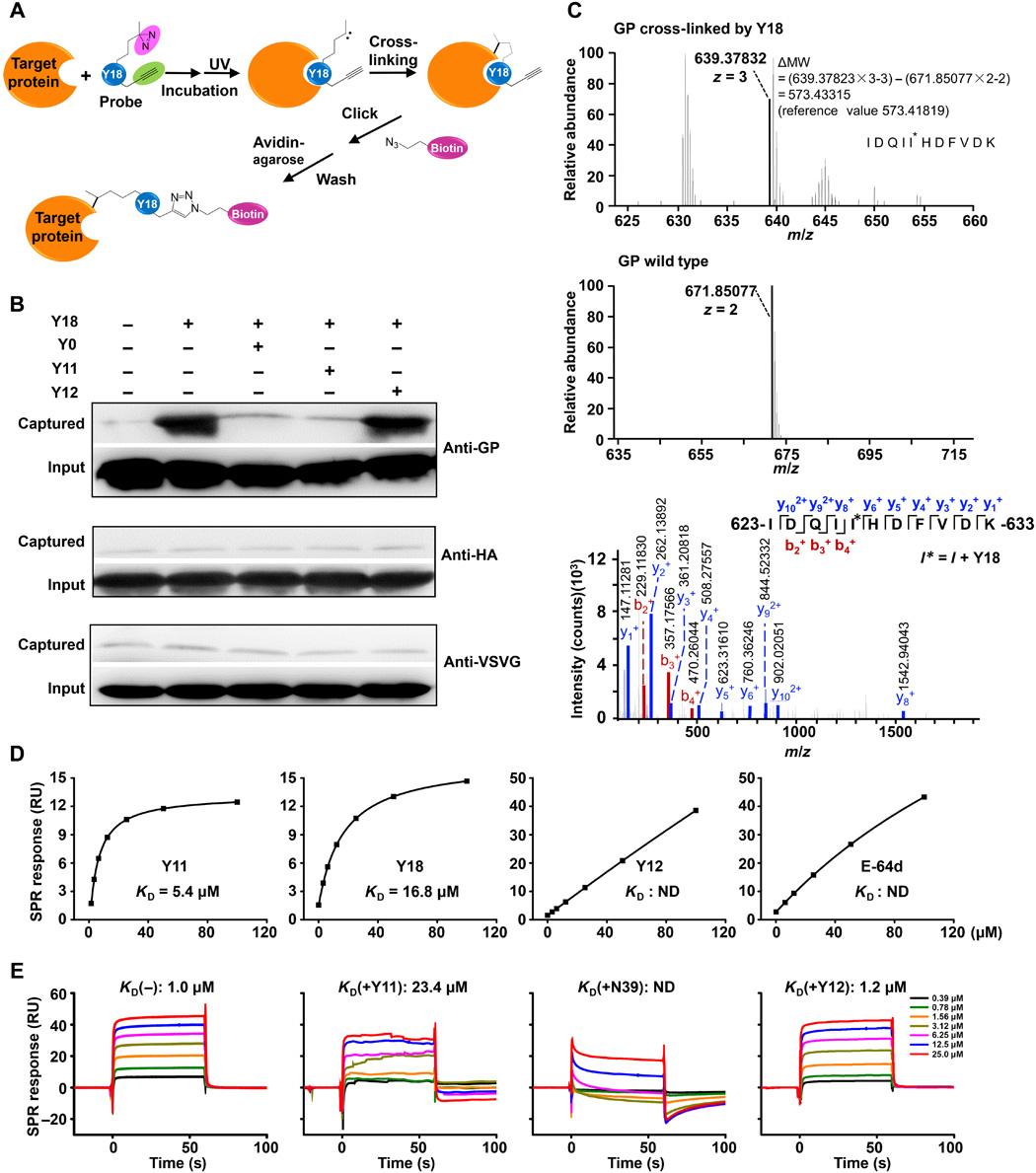
A trimer-of-hairpins motif has been identified in triggering virus-cell fusion within a variety of viral envelopes. Chemically manipulating such a motif represents current repertoire of viral fusion inhibitors. Here, we report that triterpenoids, a class of natural products, antagonize this trimer-of-hairpins via its constitutive heptad repeat-2 (HR2), a prevalent α-helical coil in class I viral fusion proteins. Triterpenoids inhibit the entry of Ebola, Marburg, HIV, and influenza A viruses with distinct structure-activity relationships. Specifically, triterpenoid probes capture the viral envelope via photocrosslinking HR2. Profiling the Ebola HR2-triterpenoid interactions using amino acid substitution, surface plasmon resonance, and nuclear magnetic resonance revealed six residues accessible to triterpenoids, leading to wrapping of the hydrophobic helix and blocking of the HR1-HR2 interaction critical in the trimer-of-hairpins formation. This finding was also observed in the envelopes of HIV and influenza A viruses and might potentially extend to a broader variety of viruses, providing a mechanistic insight into triterpenoid-mediated modulation of viral fusion.
三萜类化合物通过包裹病毒包膜中普遍存在的 HR2 结构域来操纵病毒与宿主的广泛融合
在多种病毒包膜中,已经发现了一个触发病毒-细胞融合的发夹三聚体图案。通过化学方法处理这样的基团是目前病毒融合抑制剂的主要手段。在这里,我们报告了三萜类天然产品通过构成型七联重复-2(HR2)来拮抗这种发夹三聚体,HR2 是 I 类病毒融合蛋白中常见的 α 螺旋线圈。三萜类化合物抑制埃博拉病毒、马尔堡病毒、艾滋病病毒和甲型流感病毒的进入,其结构与活性关系截然不同。具体来说,三萜类探针通过光交联 HR2 捕获病毒包膜。利用氨基酸置换、表面等离子共振和核磁共振分析埃博拉 HR2 与三萜类化合物的相互作用,发现三萜类化合物可接触六个残基,导致疏水螺旋缠绕,阻断 HR1-HR2 相互作用,这对三聚发素的形成至关重要。在艾滋病毒和甲型流感病毒的包膜中也观察到了这一发现,并有可能扩展到更多种类的病毒,为三萜类化合物介导的病毒融合调节提供了机理上的见解。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Science Advances
综合性期刊-综合性期刊
CiteScore
21.40
自引率
1.50%
发文量
1937
审稿时长
29 weeks
期刊介绍:
Science Advances, an open-access journal by AAAS, publishes impactful research in diverse scientific areas. It aims for fair, fast, and expert peer review, providing freely accessible research to readers. Led by distinguished scientists, the journal supports AAAS's mission by extending Science magazine's capacity to identify and promote significant advances. Evolving digital publishing technologies play a crucial role in advancing AAAS's global mission for science communication and benefitting humankind.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


