Improved gelling and emulsifying properties of myofibrillar protein from frozen shrimp (Litopenaeus vannamei) by high-intensity ultrasound
Abstract
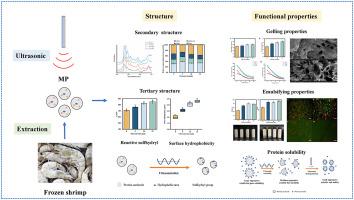
The deterioration of shrimp protein functionality during chilling and frozen storage has recently attracted wide attention due to its adverse effects on shrimp quality. The high-intensity ultrasound (20 kHz, 400 W) was applied to improve the protein structure and functional properties of frozen shrimp (Litopenaeus vannamei) in this study. Fourier transform infrared (FTIR) spectroscopy showed that the cavitation effect of ultrasound could change the secondary structure of myofibrillar protein (MP) by turning the α-helix and random coil to β-sheet and β-turn. The unfolding of MP conformation increased the reactive sulfhydryl content and surface hydrophobicity accordingly. The structure changes eventually led to the significant improvement of protein functionality. Gel properties analysis showed after ultrasonic treatment, the gel strength and water holding capacity of protein gels were increased gradually. Scanning electron microscopy (SEM) images also demonstrated that ultrasound treatment contributed to the formation of stable network structures in protein gels. In addition, ultrasonic homogenization could increase the emulsifying ability (45.54 m2/g to 78.82 m2/g) and stability of protein in a short time, which might be related to the decrease of particle size. Confocal laser scanning microscope (CLSM) analysis further illustrated that ultrasound treatment made the emulsion distribution more uniform. The application of these findings will improve the deep processing ability and expand the commercialization of frozen shrimp products.

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


