The mechanistic basis of cargo selection during Golgi maturation
IF 12.5
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
Abstract
The multiple cisternae of the Golgi apparatus contain resident membrane proteins crucial for lipid and protein glycosylation. How Golgi residents remain in their designated compartments despite a constant flow of secretory cargo is incompletely understood. Here, we determine the structure of the COPI vesicle coat containing GOLPH3, an adaptor protein that binds the cytosolic tails of many Golgi residents. Analysis of this structure, together with structure-guided mutagenesis and functional assays, reveals how GOLPH3 uses coincidence detection of COPI and lipids to engage Golgi residents preferentially at late cisternae. Our findings rationalize the logic of cisternal maturation and explain how COPI can engage different types of substrates in different Golgi cisternae to retrieve some proteins back to the ER while retaining others within the Golgi apparatus.
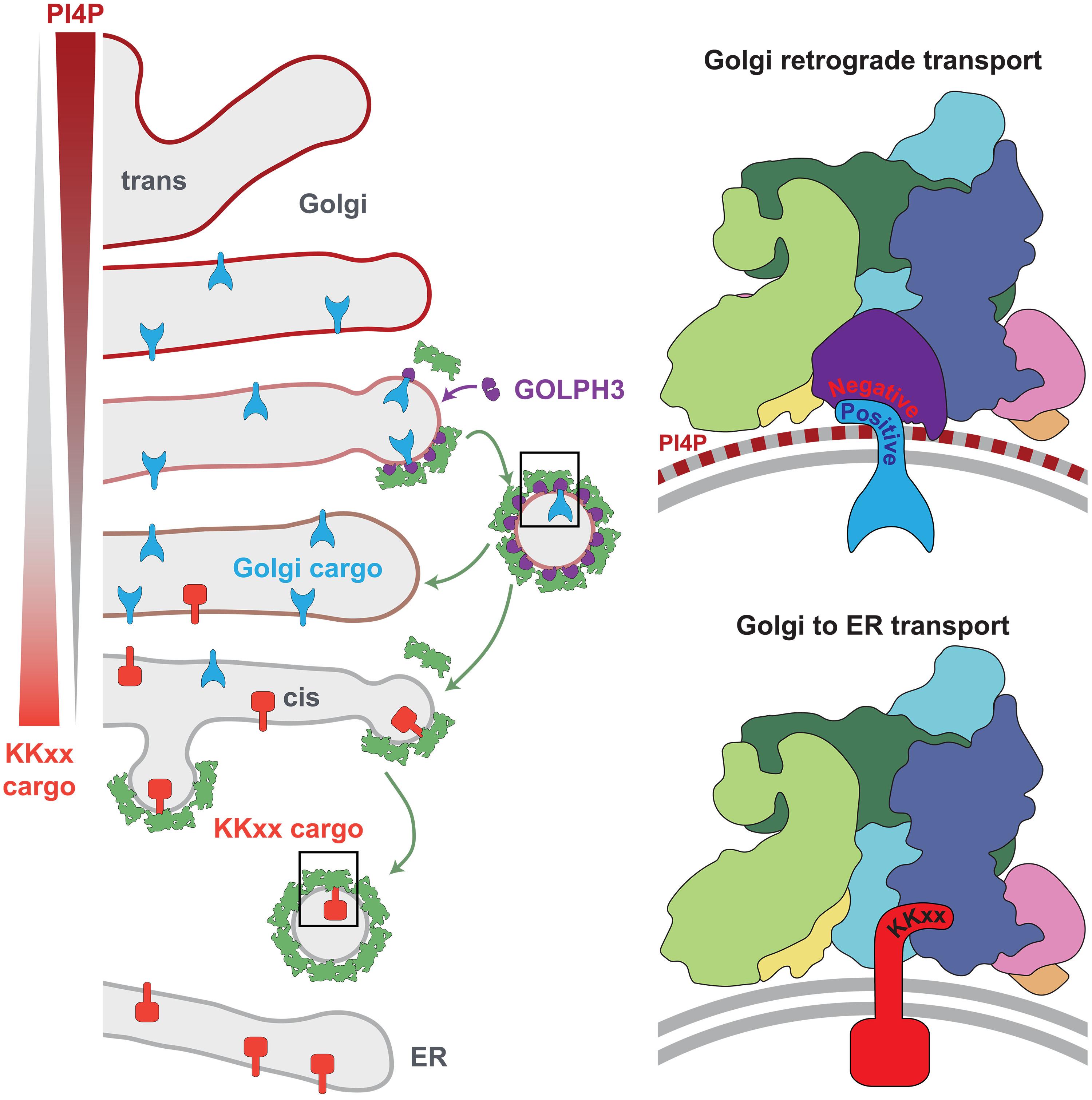
高尔基成熟过程中货物选择的机理基础
高尔基体的多个贮池含有对脂质和蛋白质糖基化至关重要的常驻膜蛋白。尽管有源源不断的分泌物,高尔基岛上的居民是如何留在他们指定的隔间里的,目前还不完全清楚。在这里,我们确定了含有GOLPH3的COPI囊泡外壳的结构,GOLPH3是一种结合许多高尔基体的细胞质尾部的连接蛋白。对该结构的分析,以及结构引导的诱变和功能分析,揭示了GOLPH3如何利用COPI和脂质的一致性检测优先参与高尔基体居民的晚期池。我们的发现合理化了池成熟的逻辑,并解释了COPI如何在不同的高尔基池中参与不同类型的底物,将一些蛋白质带回内质网,同时将其他蛋白质保留在高尔基体中。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Science Advances
综合性期刊-综合性期刊
CiteScore
21.40
自引率
1.50%
发文量
1937
审稿时长
29 weeks
期刊介绍:
Science Advances, an open-access journal by AAAS, publishes impactful research in diverse scientific areas. It aims for fair, fast, and expert peer review, providing freely accessible research to readers. Led by distinguished scientists, the journal supports AAAS's mission by extending Science magazine's capacity to identify and promote significant advances. Evolving digital publishing technologies play a crucial role in advancing AAAS's global mission for science communication and benefitting humankind.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


