Application of 1,3-disubstituted thioureas as a key synthons for the synthesis of novel 5,7-disubstituted perhydroimidazo[4,5-e][1,2,4]triazinethiones
IF 2.2
3区 化学
Q2 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
A series of 1-alkyl-3-arylureas and thioureas, a key synthons for the synthesis of various heterocycles, have been synthesized. The corresponding 4,5-dihydroxyimidzolidin-2-ones and thiones were prepared from (thio)ureas with glyoxal, and the selectivity of their reaction with thiosemicarbazide was studied. Regioisomeric 7-alkyl-5-aryl- and 5-alkyl-7-aryl-3-thioxoperhydroimidazo[4,5-e][1,2,4]triazin-6-ones(thiones) were prepared and separated by fractional crystallization. Intermediate 3-alkyl-2-(1-(4-chlorophenyl)-5-ethoxy-2-oxoimidazolidin-4-yl)hydrazinecarbothioamides were also isolated. The structures of the regioisomers and intermediates were unambiguously determined by two-dimensional NMR NOESY spectral data and X-ray diffraction studies.
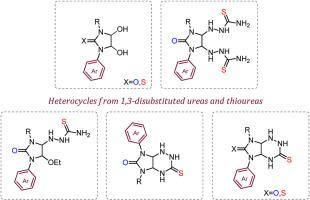
应用1,3-二取代硫脲作为合成新型5,7-二取代过氢咪唑[4,5-e][1,2,4]三嗪硫酮的关键合成物
合成了一系列的1-烷基-3-芳脲和硫脲,它们是合成各种杂环化合物的关键化合物。以(硫)脲和乙二醛为原料制备了相应的4,5-二羟基咪唑烷酮和硫酮,并研究了它们与硫脲的选择性反应。制备了区域异构体7-烷基-5-芳基-和5-烷基-7-芳基-3-硫氧过氢咪唑[4,5-e][1,2,4]三嗪-6-酮(硫酮),并采用分馏结晶法分离。还分离到了中间的3-烷基-2-(1-(4-氯苯基)-5-乙氧基-2-氧咪唑烷-4-基)肼碳硫酰胺。区域异构体和中间体的结构是通过二维核磁共振波谱数据和x射线衍射研究明确确定的。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Tetrahedron
化学-有机化学
CiteScore
3.90
自引率
4.80%
发文量
439
审稿时长
34 days
期刊介绍:
Tetrahedron publishes full accounts of research having outstanding significance in the broad field of organic chemistry and its related disciplines, such as organic materials and bio-organic chemistry.
Regular papers in Tetrahedron are expected to represent detailed accounts of an original study having substantially greater scope and details than that found in a communication, as published in Tetrahedron Letters.
Tetrahedron also publishes thematic collections of papers as special issues and ''Reports'', commissioned in-depth reviews providing a comprehensive overview of a research area.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


