Highly regio- and diastereoselective construction of spirocyclic compounds via palladium-catalyzed [3 + 2] cycloaddition of vinyl methylene cyclic carbonates with pyrrolidinones
IF 2.7
3区 化学
Q1 CHEMISTRY, ORGANIC
引用次数: 0
Abstract
A palladium-catalyzed [3 + 2] cycloaddition reaction of vinyl methylene cyclic carbonates with pyrrolidinones was developed. This protocol demonstrates remarkable versatility, enabling the facile construction of diverse [5,5]-spirolactam scaffolds in excellent yields with outstanding diastereoselectivity. The synthetic utility of this protocol was further demonstrated through productive transformations of the resulting spirolactam products, highlighting their potential as valuable intermediates for complex molecule synthesis.
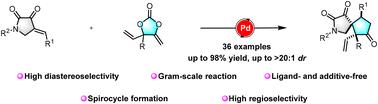
钯催化[3 + 2]环加成羰基环亚甲基碳酸酯与吡咯烷酮的高区域选择性和非对映选择性结构
研究了钯催化的亚环碳酸乙烯基与吡咯烷酮的[3 + 2]环加成反应。该方案具有显著的通用性,能够以优异的产率和出色的非对映选择性轻松构建多种[5,5]-螺内酰胺支架。通过所得到的螺内酰胺产品的生产性转化,进一步证明了该方案的合成效用,突出了它们作为复杂分子合成有价值的中间体的潜力。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Organic & Biomolecular Chemistry
化学-有机化学
CiteScore
5.50
自引率
9.40%
发文量
1056
审稿时长
1.3 months
期刊介绍:
Organic & Biomolecular Chemistry is an international journal using integrated research in chemistry-organic chemistry. Founded in 2003 by the Royal Society of Chemistry, the journal is published in Semimonthly issues and has been indexed by SCIE, a leading international database. The journal focuses on the key research and cutting-edge progress in the field of chemistry-organic chemistry, publishes and reports the research results in this field in a timely manner, and is committed to becoming a window and platform for rapid academic exchanges among peers in this field. The journal's impact factor in 2023 is 2.9, and its CiteScore is 5.5.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


