Switchable skeletal editing of quinolines enabled by cyclizative sequential rearrangements
IF 20.2
1区 化学
Q1 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
The rapid diversification of core ring structures in complex molecules through switchable skeletal editing is valuable in the drug discovery process. However, controllable methods for chemically divergent modifications of azaarene frameworks using common substrates are challenging, despite the potential to maximize structural diversity and complexity. Here we report the tunable skeletal editing of quinolines through Brønsted acid-catalysed multicomponent reactions of quinoline N-oxides, dialkyl acetylenedicarboxylates and water to generate nitrogen-containing heteroaromatic compounds together with linear compounds in a modular fashion. Specifically, in a one-pot procedure, after cyclization and sequential rearrangement processes, the quinoline N-oxides are easily converted into unique 2-substituted indolines. These then undergo acid-promoted fragmentation to give indoles, base-facilitated ring-opening to afford 2-alkenylanilines and oxidative cyclization to yield isoquinolinones. Catalytic asymmetric skeletal editing of quinolines is also realized, providing enantioenriched benzazepines bearing quaternary stereocentres, and late-stage skeletal modification of quinoline cores in several drugs is demonstrated. Switchable skeletal editing is valuable in drug discovery, but controllable methods for chemodivergent modifications of azaarenes are challenging. Now transformations of quinolines have been achieved by leveraging cyclizative rearrangement strategies, enabling the construction of four different frameworks, including the enantioselective skeletal editing of quinolines into benzazepines.
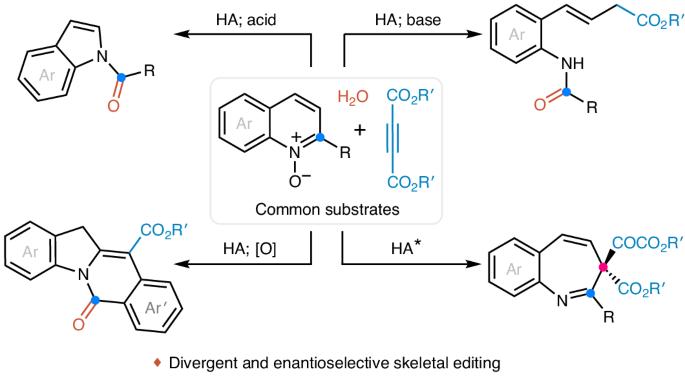
通过循环顺序重排实现喹啉类的可切换骨架编辑。
通过可切换的骨架编辑在复杂分子中快速多样化核心环结构在药物发现过程中是有价值的。然而,尽管可以最大限度地提高结构多样性和复杂性,但使用普通底物对氮杂芳烃框架进行化学差异性修饰的可控方法仍然具有挑战性。在这里,我们报道了喹啉的可调骨架编辑,通过Brønsted酸催化喹啉n-氧化物、二烷基乙炔二羧酸盐和水的多组分反应,以模块化的方式生成含氮杂芳香化合物和线性化合物。具体来说,在一锅过程中,经过环化和顺序重排过程,喹啉n -氧化物很容易转化为独特的2-取代吲哚。然后经过酸促进裂解生成吲哚,碱促进开环生成2-肯苯胺,氧化环化生成异喹啉酮。喹啉类药物的催化不对称骨架编辑也得以实现,提供了具有四元立体中心的对映体富集的苯氮卓类药物,并证明了几种药物中喹啉核心的后期骨架修饰。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature chemistry
化学-化学综合
CiteScore
29.60
自引率
1.40%
发文量
226
审稿时长
1.7 months
期刊介绍:
Nature Chemistry is a monthly journal that publishes groundbreaking and significant research in all areas of chemistry. It covers traditional subjects such as analytical, inorganic, organic, and physical chemistry, as well as a wide range of other topics including catalysis, computational and theoretical chemistry, and environmental chemistry.
The journal also features interdisciplinary research at the interface of chemistry with biology, materials science, nanotechnology, and physics. Manuscripts detailing such multidisciplinary work are encouraged, as long as the central theme pertains to chemistry.
Aside from primary research, Nature Chemistry publishes review articles, news and views, research highlights from other journals, commentaries, book reviews, correspondence, and analysis of the broader chemical landscape. It also addresses crucial issues related to education, funding, policy, intellectual property, and the societal impact of chemistry.
Nature Chemistry is dedicated to ensuring the highest standards of original research through a fair and rigorous review process. It offers authors maximum visibility for their papers, access to a broad readership, exceptional copy editing and production standards, rapid publication, and independence from academic societies and other vested interests.
Overall, Nature Chemistry aims to be the authoritative voice of the global chemical community.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


