Effect of lithium doping on the electrochemical properties of Bi2Sr2Co2Oy misfit-layered oxides for supercapacitor applications
IF 5.6
3区 材料科学
Q1 ELECTROCHEMISTRY
引用次数: 0
Abstract
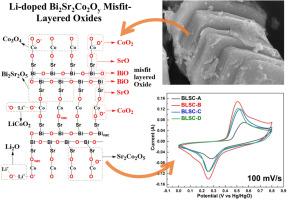
The effect of lithium (Li) doping on Bi2Sr2Co2Oy misfit layer oxides for supercapacitor applications is reported for the first time in this research paper. Four different compositions, which included Bi2Li0.1Sr1.9Co2Oy, Bi2Li0.1Sr2Co1.9Oy, Bi2Li0.2Sr1.8Co2Oy, and Bi2Li0.2Sr2Co1.8Oy, were synthesized using a solution combustion method. The obtained powders were further calcined at 900 °C for 10 hours and subsequently characterized using field emission scanning electron microscopy (FE-SEM), energy-dispersive X-ray spectroscopy (EDX), X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), Brunauer-Emmett-Teller (BET) analysis and Raman spectroscopy to study the morphology, crystal structure, surface area, elemental identification, quantification, and bonding nature of Li-doped Bi2Sr2Co2Oy. Cyclic voltammetry (CV), galvanostatic charge-discharge (GCD), and electrochemical impedance spectroscopy (EIS) were performed for electrochemical characterization to study the effect of Li doping on the charge storage performance of Bi2Sr2Co2Oy. Li doping enhanced the charge storage ability of Bi2Sr2Co2Oy by 3 to 5 fold, and all compositions demonstrated a diffusion-based charge storage mechanism. The Bi2Li0.1Sr2Co1.9Oy solid-state asymmetric device attained the highest specific capacity of 299.6 C/g at 5 mV/s and 241.5 C/g at 1 A/g with 53.7 Wh/kg energy density.

锂掺杂对超级电容器用Bi2 Sr2 Co2 Oy错配层氧化物电化学性能的影响
本文首次报道了锂掺杂对超级电容器用Bi2Sr2Co2Oy失配层氧化物的影响。采用溶液燃烧法合成了Bi2Li0.1Sr1.9Co2Oy、Bi2Li0.1Sr2Co1.9Oy、Bi2Li0.2Sr1.8Co2Oy和Bi2Li0.2Sr2Co1.8Oy四种不同的化合物。得到的粉末在900°C下进一步煅烧10小时,随后使用场发射扫描电镜(FE-SEM),能量色散x射线能谱(EDX), x射线衍射(XRD), x射线光电子能谱(XPS), brunauer - emmet - teller (BET)分析和拉曼光谱进行表征,研究li掺杂Bi2Sr2Co2Oy的形貌,晶体结构,表面积,元素鉴定,定量和键合性质。采用循环伏安法(CV)、恒流充放电法(GCD)和电化学阻抗谱法(EIS)进行电化学表征,研究Li掺杂对Bi2Sr2Co2Oy电荷存储性能的影响。Li掺杂使Bi2Sr2Co2Oy的电荷存储能力提高了3 ~ 5倍,且各组分均表现出基于扩散的电荷存储机制。Bi2Li0.1Sr2Co1.9Oy固态非对称器件在5mv /s时获得最高比容量299.6 C/g,在1a /g时获得最高比容量241.5 C/g,能量密度为53.7 Wh/kg。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Electrochimica Acta
工程技术-电化学
CiteScore
11.30
自引率
6.10%
发文量
1634
审稿时长
41 days
期刊介绍:
Electrochimica Acta is an international journal. It is intended for the publication of both original work and reviews in the field of electrochemistry. Electrochemistry should be interpreted to mean any of the research fields covered by the Divisions of the International Society of Electrochemistry listed below, as well as emerging scientific domains covered by ISE New Topics Committee.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


