Oxidation of retromer complex controls mitochondrial translation
IF 48.5
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
Abstract
Reactive oxygen species (ROS) underlie human pathologies including cancer and neurodegeneration1,2. However, the proteins that sense ROS levels and regulate their production through their cysteine residues remain ill defined. Here, using systematic base-editing and computational screens, we identify cysteines in VPS35, a member of the retromer trafficking complex3, that phenocopy inhibition of mitochondrial translation when mutated. We find that VPS35 underlies a reactive metabolite-sensing pathway that lowers mitochondrial translation to decrease ROS levels. Intracellular hydrogen peroxide oxidizes cysteine residues in VPS35, resulting in retromer dissociation from endosomal membranes and subsequent plasma membrane remodelling. We demonstrate that plasma membrane localization of the retromer substrate SLC7A1 is required to sustain mitochondrial translation. Furthermore, decreasing VPS35 levels or oxidation of its ROS-sensing cysteines confers resistance to ROS-generating chemotherapies, including cisplatin, in ovarian cancer models. Thus, we identify that intracellular ROS levels are communicated to the plasma membrane through VPS35 to regulate mitochondrial translation, connecting cytosolic ROS sensing to mitochondrial ROS production. Systematic base-editing and computational screens identify specific cysteine residues on VPS35 in the retromer complex as key sensors that decrease mitochondrial translation in response to reactive oxygen species signals.
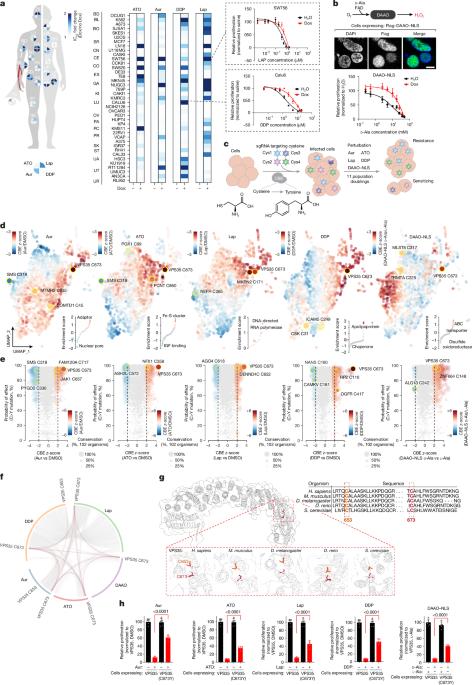

反转录复合体的氧化控制线粒体翻译
活性氧(ROS)是包括癌症和神经变性在内的人类病理的基础1,2。然而,感知ROS水平并通过其半胱氨酸残基调节其产生的蛋白质仍然不明确。在这里,使用系统的碱基编辑和计算筛选,我们鉴定了VPS35中的半胱氨酸,VPS35是逆转录物运输复合体3的成员,当线粒体翻译发生突变时,其表型抑制作用。我们发现VPS35是一个反应性代谢物感应途径的基础,该途径可以降低线粒体翻译以降低ROS水平。细胞内过氧化氢氧化VPS35中的半胱氨酸残基,导致内体膜的反转录解离和随后的质膜重构。我们证明了逆转录底物SLC7A1的质膜定位是维持线粒体翻译所必需的。此外,在卵巢癌模型中,VPS35水平的降低或其ros敏感半胱氨酸的氧化使其对产生ros的化疗(包括顺铂)产生耐药性。因此,我们发现细胞内ROS水平通过VPS35传递到质膜,调节线粒体翻译,将细胞质内ROS感知与线粒体ROS产生联系起来。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature
综合性期刊-综合性期刊
CiteScore
90.00
自引率
1.20%
发文量
3652
审稿时长
3 months
期刊介绍:
Nature is a prestigious international journal that publishes peer-reviewed research in various scientific and technological fields. The selection of articles is based on criteria such as originality, importance, interdisciplinary relevance, timeliness, accessibility, elegance, and surprising conclusions. In addition to showcasing significant scientific advances, Nature delivers rapid, authoritative, insightful news, and interpretation of current and upcoming trends impacting science, scientists, and the broader public. The journal serves a dual purpose: firstly, to promptly share noteworthy scientific advances and foster discussions among scientists, and secondly, to ensure the swift dissemination of scientific results globally, emphasizing their significance for knowledge, culture, and daily life.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


