Advancing metallomimetic catalysis through structural constraints of cationic PIII species
IF 4.2
2区 化学
Q2 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
In recent years, the concept of structural constraints on the main-group (MG) centers has emerged as a powerful strategy to enhance their reactivity. Among these, structurally constrained (SC) phosphorus centers have garnered significant attention due to their ability to cycle between two stable oxidation states, P(iii) and P(v), making them highly promising for small molecule activation and catalysis. Structural constraints grant phosphorus centers transition metal (TM)-like reactivity, enabling the activation of small molecules by these SC P(iii) centers, a reactivity previously inaccessible with conventional phosphines or other phosphorus derivatives. This feature article reviews recent advances in the chemistry of cationic, structurally constrained P(iii) (CSCP) compounds, emphasizing their ability to mimic TM behavior in small-molecule activation and catalysis, particularly through the key elementary steps of TM-based catalysis, such as oxidative addition (OA), migratory insertion (MI), ligand metathesis (LM), reductive elimination (RE), etc. The development of these SC cationic P(iii) species highlights the interplay between structural constraints and cationic charge, facilitating analogous metallomimetic reactivity in other main-group elements.
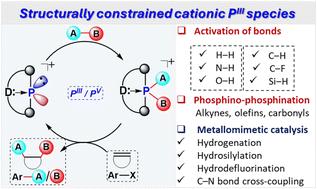
利用阳离子PIII的结构约束推进金属模拟催化。
近年来,对主群(MG)中心进行结构约束的概念已成为增强其反应性的有力策略。其中,结构约束(SC)磷中心因其在两种稳定氧化态(P(III)和P(V))之间循环的能力而引起了极大的关注,这使得它们在小分子活化和催化方面具有很大的前景。结构限制赋予磷中心类似过渡金属(TM)的反应性,使这些SC P(III)中心能够激活小分子,这是传统磷化氢或其他磷衍生物以前无法达到的反应性。本文综述了阳离子、结构受限的P(III) (CSCP)化合物的化学研究进展,强调了它们在小分子活化和催化中模仿TM行为的能力,特别是通过TM催化的关键基本步骤,如氧化加成(OA)、迁移插入(MI)、配体复分解(LM)、还原消除(RE)等。这些SC阳离子P(III)物种的发展突出了结构约束和阳离子电荷之间的相互作用,促进了其他主族元素的类似金属模拟反应性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Chemical Communications
化学-化学综合
CiteScore
8.60
自引率
4.10%
发文量
2705
审稿时长
1.4 months
期刊介绍:
ChemComm (Chemical Communications) is renowned as the fastest publisher of articles providing information on new avenues of research, drawn from all the world''s major areas of chemical research.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


