Nociceptor neurons promote PDAC progression and cancer pain by interaction with cancer-associated fibroblasts and suppression of natural killer cells
IF 25.9
1区 生物学
Q1 CELL BIOLOGY
引用次数: 0
Abstract
The emerging field of cancer neuroscience has demonstrated great progress in revealing the crucial role of the nervous system in cancer initiation and progression. Pancreatic ductal adenocarcinoma (PDAC) is characterized by perineural invasion and modulated by autonomic (sympathetic and parasympathetic) and sensory innervations. Here, we further demonstrated that within the tumor microenvironment of PDAC, nociceptor neurons interacted with cancer-associated fibroblasts (CAFs) through calcitonin gene-related peptide (CGRP) and nerve growth factor (NGF). This interaction led to the inhibition of interleukin-15 expression in CAFs, suppressing the infiltration and cytotoxic function of natural killer (NK) cells and thereby promoting PDAC progression and cancer pain. In PDAC patients, nociceptive innervation of tumor tissue is negatively correlated with the infiltration of NK cells while positively correlated with pain intensity. This association serves as an independent prognostic factor for both overall survival and relapse-free survival for PDAC patients. Our findings highlight the crucial regulation of NK cells by nociceptor neurons through interaction with CAFs in the development of PDAC. We also propose that targeting nociceptor neurons or CGRP signaling may offer a promising therapy for PDAC and cancer pain.
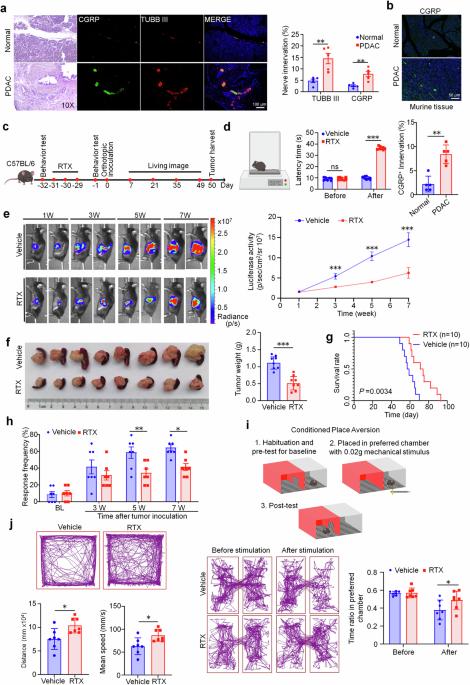

痛觉感受器神经元通过与癌症相关成纤维细胞和抑制自然杀伤细胞的相互作用促进PDAC的进展和癌症疼痛
新兴的癌症神经科学领域在揭示神经系统在癌症发生和发展中的关键作用方面取得了巨大进展。胰腺导管腺癌(PDAC)的特点是神经周围受侵,并受自主神经(交感神经和副交感神经)和感觉神经支配。在这里,我们进一步证实,在 PDAC 的肿瘤微环境中,痛觉神经元通过降钙素基因相关肽(CGRP)和神经生长因子(NGF)与癌相关成纤维细胞(CAFs)相互作用。这种相互作用抑制了 CAFs 中白细胞介素-15 的表达,抑制了自然杀伤(NK)细胞的浸润和细胞毒性功能,从而促进了 PDAC 的进展和癌痛。在 PDAC 患者中,肿瘤组织的痛觉支配与 NK 细胞的浸润呈负相关,而与疼痛强度呈正相关。这种关联是 PDAC 患者总生存期和无复发生存期的独立预后因素。我们的研究结果突出表明,在 PDAC 的发展过程中,痛觉神经元通过与 CAFs 相互作用对 NK 细胞进行关键性调控。我们还提出,以痛觉神经元或 CGRP 信号为靶点可能会为 PDAC 和癌痛提供一种有前景的疗法。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Cell Research
生物-细胞生物学
CiteScore
53.90
自引率
0.70%
发文量
2420
审稿时长
2.3 months
期刊介绍:
Cell Research (CR) is an international journal published by Springer Nature in partnership with the Center for Excellence in Molecular Cell Science, Chinese Academy of Sciences (CAS). It focuses on publishing original research articles and reviews in various areas of life sciences, particularly those related to molecular and cell biology. The journal covers a broad range of topics including cell growth, differentiation, and apoptosis; signal transduction; stem cell biology and development; chromatin, epigenetics, and transcription; RNA biology; structural and molecular biology; cancer biology and metabolism; immunity and molecular pathogenesis; molecular and cellular neuroscience; plant molecular and cell biology; and omics, system biology, and synthetic biology. CR is recognized as China's best international journal in life sciences and is part of Springer Nature's prestigious family of Molecular Cell Biology journals.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


