Transient silencing of hypermutation preserves B cell affinity during clonal bursting
IF 48.5
1区 综合性期刊
Q1 MULTIDISCIPLINARY SCIENCES
引用次数: 0
Abstract
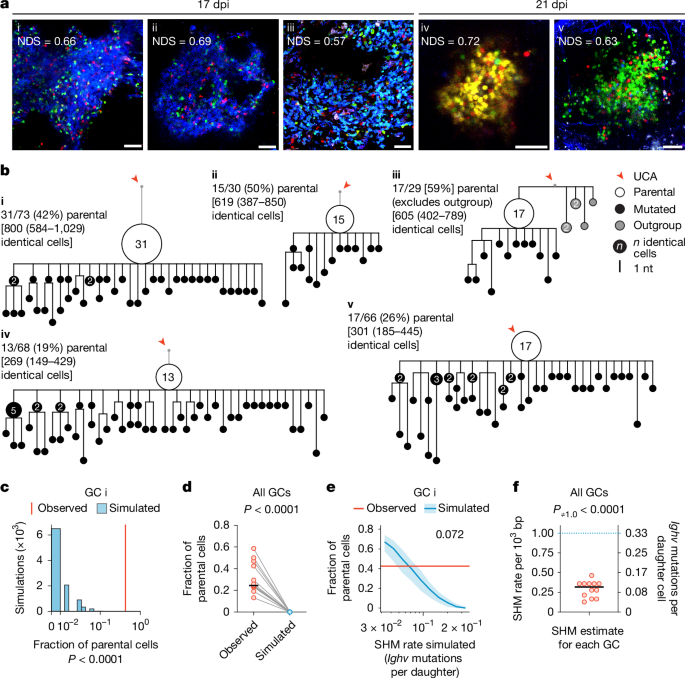
In the course of antibody affinity maturation, germinal centre (GC) B cells mutate their immunoglobulin heavy- and light-chain genes in a process known as somatic hypermutation (SHM)1–4. Panels of mutant B cells with different binding affinities for antigens are then selected in a Darwinian manner, which leads to a progressive increase in affinity among the population5. As with any Darwinian process, rare gain-of-fitness mutations must be identified and common loss-of-fitness mutations avoided6. Progressive acquisition of mutations therefore poses a risk during large proliferative bursts7, when GC B cells undergo several cell cycles in the absence of affinity-based selection8–13. Using a combination of in vivo mouse experiments and mathematical modelling, here we show that GCs achieve this balance by strongly suppressing SHM during clonal-burst-type expansion, so that a large fraction of the progeny generated by these bursts does not deviate from their ancestral genotype. Intravital imaging and image-based cell sorting of a mouse strain carrying a reporter of cyclin-dependent kinase 2 (CDK2) activity showed that B cells that are actively undergoing proliferative bursts lack the transient CDK2low ‘G0-like’ phase of the cell cycle in which SHM takes place. We propose a model in which inertially cycling B cells mostly delay SHM until the G0-like phase that follows their final round of division in the GC dark zone, thus maintaining affinity as they clonally expand in the absence of selection. B cell germinal centres achieve a balance between clonal expansion and sequence diversification by suppressing somatic hypermutation during proliferative clonal bursts.

在克隆破裂期间,超突变的短暂沉默保留了B细胞的亲和力
在抗体亲和成熟过程中,生发中心(GC) B细胞在称为体细胞超突变(SHM)1,2,3,4的过程中突变其免疫球蛋白重链和轻链基因。然后以达尔文的方式选择具有不同抗原结合亲和力的突变B细胞组,这导致群体之间的亲和力逐渐增加5。与任何达尔文过程一样,罕见的适应性突变必须被识别出来,而常见的适应性突变必须被避免。因此,当GC B细胞在缺乏亲和选择的情况下经历几个细胞周期8,9,10,11,12,13时,突变的渐进式获取在大规模增殖爆发中存在风险。通过结合小鼠体内实验和数学模型,我们发现GCs通过在克隆爆发型扩增过程中强烈抑制SHM来实现这种平衡,因此这些爆发产生的大部分后代不会偏离其祖先的基因型。一种携带周期蛋白依赖性激酶2 (CDK2)活性报告基因的小鼠品系的活体成像和基于图像的细胞分选显示,积极经历增殖爆发的B细胞缺乏发生SHM的细胞周期的瞬时CDK2low“g0样”期。我们提出了一个模型,在这个模型中,惯性循环的B细胞大多将SHM延迟到GC暗区最后一轮分裂后的g0样期,从而在没有选择的情况下保持亲和性。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文
来源期刊

Nature
综合性期刊-综合性期刊
CiteScore
90.00
自引率
1.20%
发文量
3652
审稿时长
3 months
期刊介绍:
Nature is a prestigious international journal that publishes peer-reviewed research in various scientific and technological fields. The selection of articles is based on criteria such as originality, importance, interdisciplinary relevance, timeliness, accessibility, elegance, and surprising conclusions. In addition to showcasing significant scientific advances, Nature delivers rapid, authoritative, insightful news, and interpretation of current and upcoming trends impacting science, scientists, and the broader public. The journal serves a dual purpose: firstly, to promptly share noteworthy scientific advances and foster discussions among scientists, and secondly, to ensure the swift dissemination of scientific results globally, emphasizing their significance for knowledge, culture, and daily life.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


