The Role of Kinetic Asymmetry in Chemical and Thermodynamic Coupling
IF 3.1
Q2 CHEMISTRY, MULTIDISCIPLINARY
引用次数: 0
Abstract
The input of energy can shift an isomerization reaction A⇌B away from equilibrium, but which way, in favor of A or in favor of B? The answer to this question lies in understanding kinetic asymmetry, a concept first discussed in the context of how energy from an oscillating or fluctuating perturbation can act in concert with a catalyst to drive a reaction away from equilibrium. The key theoretical result is the non-equilibrium pumping equality that generalizes the idea of the equilibrium constant to the non-equilibrium steady-state.


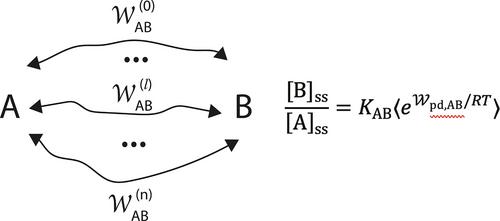
动力学不对称在化学和热力学耦合中的作用
输入的能量可以使异构化反应A + B偏离平衡,但是哪一个方向,是偏向A还是偏向B?这个问题的答案在于理解动力学不对称,动力学不对称这个概念最初是在振荡或波动扰动的能量如何与催化剂协同作用以使反应偏离平衡的背景下讨论的。关键的理论结果是非平衡泵送方程,它将平衡常数的概念推广到非平衡稳态。
本文章由计算机程序翻译,如有差异,请以英文原文为准。
求助全文
约1分钟内获得全文
求助全文

 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


