Unraveling the Roles of the ZnO Surface Structure and Second Metal Doping in Tuning the Catalytic Performance of Ethane Dehydrogenation
IF 11.3
1区 化学
Q1 CHEMISTRY, PHYSICAL
引用次数: 0
Abstract
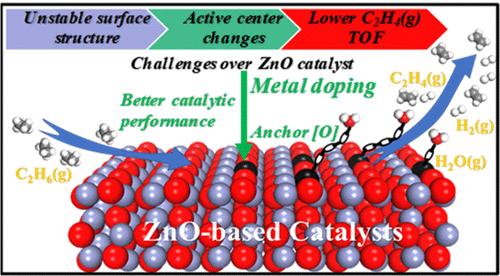
The ZnO surface is easily reduced during alkane dehydrogenation owing to the formation of surface hydrogen species, resulting in poor catalytic performance. Aiming at revealing ZnO surface structure evolution, the degree of surface reduction, catalyst stability, and the type of key species contributing to surface reduction in the ethane dehydrogenation (EDH) reaction, this work fully investigated the mechanism of the EDH reaction over ZnO and a series of ZnO-based catalysts by using DFT calculations and kMC simulations. The results show that ZnO surface reduction is mainly caused by the interaction of surface H* species from EDH with surface lattice oxygen to generate H2O(g), leading to surface oxygen vacancy (Ov) formation over ZnO. As the EDH reaction proceeds, the number of Ov increases, and the active center gradually shifts from the Zn–O site to the Zn–Zncus site, decreasing the C2H4(g) formation activity and ultimately deactivating the ZnO catalyst. Furthermore, the second metal M is introduced into the ZnO surface to construct M/ZnO catalysts, and the Mn/ZnO catalyst is screened out to present better catalytic performance, which is not easily reduced. This work is of great significance in laying a solid foundation for optimizing the catalytic performance of the EDH reaction over ZnO-based catalysts.
求助全文
约1分钟内获得全文
求助全文
来源期刊

ACS Catalysis
CHEMISTRY, PHYSICAL-
CiteScore
20.80
自引率
6.20%
发文量
1253
审稿时长
1.5 months
期刊介绍:
ACS Catalysis is an esteemed journal that publishes original research in the fields of heterogeneous catalysis, molecular catalysis, and biocatalysis. It offers broad coverage across diverse areas such as life sciences, organometallics and synthesis, photochemistry and electrochemistry, drug discovery and synthesis, materials science, environmental protection, polymer discovery and synthesis, and energy and fuels.
The scope of the journal is to showcase innovative work in various aspects of catalysis. This includes new reactions and novel synthetic approaches utilizing known catalysts, the discovery or modification of new catalysts, elucidation of catalytic mechanisms through cutting-edge investigations, practical enhancements of existing processes, as well as conceptual advances in the field. Contributions to ACS Catalysis can encompass both experimental and theoretical research focused on catalytic molecules, macromolecules, and materials that exhibit catalytic turnover.
 求助内容:
求助内容: 应助结果提醒方式:
应助结果提醒方式:


